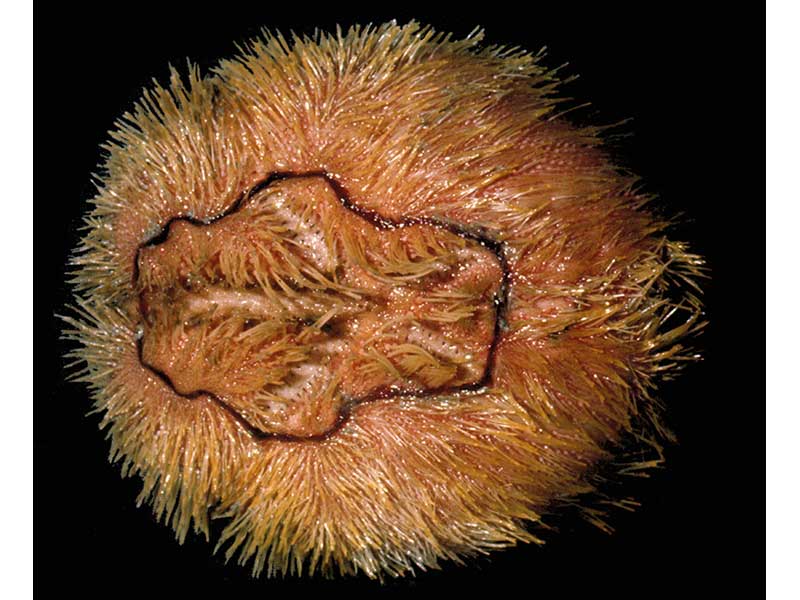
Spiny mudlark (Brissopsis lyrifera)
Distribution data supplied by the Ocean Biodiversity Information System (OBIS). To interrogate UK data visit the NBN Atlas.Map Help
| Researched by | Georgina Budd | Refereed by | Dr Karin Hollertz |
| Authority | (Forbes, 1841) | ||
| Other common names | - | Synonyms | - |
Summary
Description
A reddish-brown coloured heart urchin that is longer (7 cm) than wide. The 'test' (calcareous skeletal plates, joined together to form a complete shell) is covered in mobile spines, which are coarser and sparser in comparison with Echinocardium species. Particularly characteristic of Brissopsis lyrifera is a narrow band of ciliated dark spines which rings all five ambulacra petals on the upper surface. It is the only heart urchin likely to be found in muddy sediments.
Recorded distribution in Britain and Ireland
Recorded off the west, north and east coasts of the British Isles, but not off the south coast. Common in deep water.Global distribution
Brissopsis lyrifera may be found in offshore or inshore stable sediments from Norway and Iceland to South Africa and the Mediterranean. Also present on the east coast of North America but not Greenland.Habitat
Brissopsis lyrifera lives buried in fine mud or muddy sands offshore and in shallower, stable near shore environments.Depth range
5-365 mIdentifying features
- A rather flattened, irregular shaped urchin with a bilateral symmetry superimposed on the basic radial plan of five skeletal plates that form the 'test'.
- The 'test' is generally slightly longer than wide and distinctly notched on its anterior margin.
- In profile, the highest point of the 'test' is towards the posterior.
- The ambulacra are deeply recessed and form distinctive petal shaped areas on the upper surface.
- A petalliferous fasicole (conspicuous petal-shaped ring) of ciliated spines encloses the ambulacral petals.
- The anus is outside the apical system on the posterior edge of the 'test' and is surrounded by ciliated spines, termed the 'sub-anal fasicole'.
- The mouth is found towards the front edge of the lower surface and lacks the complex dental apparatus found in some urchin groups.
Additional information
-none-Listed by
- none -
Biology review
Taxonomy
| Level | Scientific name | Common name |
|---|---|---|
| Phylum | Echinodermata | Starfish, brittlestars, sea urchins & sea cucumbers |
| Class | Echinoidea | Sea urchins, heart urchins and sand dollars |
| Order | Spatangoida | |
| Family | Brissidae | |
| Genus | Brissopsis | |
| Authority | (Forbes, 1841) | |
| Recent Synonyms | ||
Biology
| Parameter | Data | ||
|---|---|---|---|
| Typical abundance | |||
| Male size range | <70mm | ||
| Male size at maturity | 30-60mm | ||
| Female size range | 30-60mm | ||
| Female size at maturity | |||
| Growth form | Globose | ||
| Growth rate | 10-15mm/year | ||
| Body flexibility | None (less than 10 degrees) | ||
| Mobility | Burrower | ||
| Characteristic feeding method | No information, Sub-surface deposit feeder | ||
| Diet/food source | Detritivore | ||
| Typically feeds on | Organic detritus, foraminifers and other small organisms within sediment. | ||
| Sociability | Gregarious | ||
| Environmental position | Infaunal | ||
| Dependency | No text entered. | ||
| Supports | Host Ulophysema öresundense (Brattström), an ascothoracidan parasite living in irregular sea urchins. | ||
| Is the species harmful? | No | ||
Biology information
Size. Ferrand et al. (1988), studied Brissopsis lyrifera in the Gulf of Lions, Mediterranean Sea and found the 'test' length to be unrelated to the sex of the animal.
Feeding. Brissopsis lyrifera are capable of both deposit-feeding and filter-feeding although ventilation rates are not high enough to sustain the animal on filter-feeding alone (Hollertz, 2002). Brissopsis lyrifera is reported to feed selectively on carbon- and nitrogen-rich particles (Hollertz, 2002). Mucus trapping of fine-organic rich particles and selective collection by the sticky tube feet around the mouth is thought to be the mechanisms underlying this selectivity (Hollertz, 2002). Characteristically, Brissopsis lyrifera is a sub-surface deposit feeder, but Hollertz (1998) observed Brissopsis lyrifera to emerge from the sediment and feed close to the surface in response to the addition of organic matter. When buried in the sediment, Brissopsis lyrifera maintains contact with the surface through a funnel. The funnel is kept clear of sediment by tube feet that also line the funnel with mucus (Hollertz, 2002).
In the spatangoids, to which Brissopsis lyrifera belongs, the tube foot pattern typical of the urchins is drastically altered, owing to its highly developed burrowing habit. In addition, the suckered tube feet are no longer required and they are functionally replaced (in appropriate positions) by burrow-building, sensory or feeding tube feet. Feeding is achieved by a combination of ciliary action and the action of these sticky, feeding tube feet. The feeding tube feet pass material from the substratum into the mouth where organic matter adhering to it is digested and the particular matter voided via the anus (Nichols, 1969).
Population densities. Brissopsis lyrifera is a gregarious species. Tunberg (1991) found densities of Brissopsis lyrifera to be up to 30 individuals per m² at various locations along the Swedish coast. However, in the North Sea densities of up to 60 individuals per m² have been reported (Ursin, 1960).
Mobility and burrowing. Despite being a conspicuous and large animal, very little is known about the natural burrowing behaviour of Brissopsis lyrifera, e.g. how much time it spends on the surface and how fast it moves. It is known to burrow about 2 cm below the surface of the substratum and, due to the fact that it moves with a rocking motion through the sediment, is capable of reworking relatively large volumes of sediment (Hollertz & Duchêne, 2001). In laboratory conditions, Hollertz (1998) calculated the locomotion rate of Brissopsis lyrifera to be 11 mm/hr, and, using a formula given by Schinner (1993), estimated the turnover rate of sediment by Brissopsis lyrifera to be 8.0 cm² per hour. Further research by Hollertz & Duchêne, (2001) found that Brissopsis lyrifera reworked between 14 and 22 ml of sediment per hour depending on temperature. The temperature was found to affect burrowing activity significantly and it was almost doubled when temperature was raised from 7 to 14 °C. The burrowing activity has a pronounced effect on the surrounding sediment by increasing the oxygen concentration in the sediment and thus stimulating the growth of microorganisms and decomposition of organic material (Hollertz & Duchêne, 2001).
Habitat preferences
| Parameter | Data |
|---|---|
| Physiographic preferences | Open coast, Offshore seabed, Sea loch or Sea lough |
| Biological zone preferences | Bathybenthic (Bathyal), Circalittoral offshore, Lower circalittoral |
| Substratum / habitat preferences | Mud, Muddy sand |
| Tidal strength preferences | Very weak (negligible), Weak < 1 knot (<0.5 m/sec.) |
| Wave exposure preferences | Not relevant |
| Salinity preferences | Full (30-40 psu), Variable (18-40 psu) |
| Depth range | 5-365 m |
| Other preferences | No text entered |
| Migration Pattern | Non-migratory or resident |
Habitat Information
Brissopsis lyrifera typically co-occurs with the brittle star, Amphiura chiajei, on muddy, soft bottom areas of the North Sea, the Skagerrak and the Kattegat (Hollertz et al., 1998).Life history
Adult characteristics
| Parameter | Data |
|---|---|
| Reproductive type | Gonochoristic (dioecious) |
| Reproductive frequency | Semelparous or monotely |
| Fecundity (number of eggs) | >1,000,000 |
| Generation time | See additional information |
| Age at maturity | 4 years |
| Season | Summer - Autumn |
| Life span | See additional information |
Larval characteristics
| Parameter | Data |
|---|---|
| Larval/propagule type | - |
| Larval/juvenile development | Planktotrophic |
| Duration of larval stage | See additional information |
| Larval dispersal potential | Greater than 10 km |
| Larval settlement period | Insufficient information |
Life history information
Reproduction and generation time. Echinoids have separate sexes. Fertilization is external and the majority have a free-swimming pelagic larva (Fish & Fish, 1996).
From observations made along the Northumbrian coast, Buchanan (1967), described Brissopsis lyrifera as a highly productive, short-lived but fast-growing species. The population he studied showed clear evidence of successful and consecutive annual recruitment. Specimens became sexually mature when 'test' length was >60 mm, they spawned in the summer towards the end of their 4th year and died shortly afterwards. No individuals were observed to survive to breed for a second time.
Larval settling time. Adults of Brissopsis lyrifera are burrowers, so the larval phase is the main dispersive mechanism of the urchin. Echinoderm larvae undergo a complicated and protracted metamorphosis. For instance, the larvae of other echinoderms, Echinocardium cordatum and Echinus esculentus remain in the plankton for 40 and 46-60 days respectively (Kashenko, 1994; MacBride, 1914). Thus the larvae of Brissopsis lyrifera probably remain in the plankton for a sufficient length of time to be swept away from the location of spawning to new areas or to restock existing areas (Nichols, 1969).
Sensitivity review
The MarLIN sensitivity assessment approach used below has been superseded by the MarESA (Marine Evidence-based Sensitivity Assessment) approach (see menu). The MarLIN approach was used for assessments from 1999-2010. The MarESA approach reflects the recent conservation imperatives and terminology and is used for sensitivity assessments from 2014 onwards.
Physical pressures
Use / to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Substratum loss [Show more]Substratum lossBenchmark. All of the substratum occupied by the species or biotope under consideration is removed. A single event is assumed for sensitivity assessment. Once the activity or event has stopped (or between regular events) suitable substratum remains or is deposited. Species or community recovery assumes that the substratum within the habitat preferences of the original species or community is present. Further details EvidenceBrissopsis lyrifera is an infaunal species burrowing to a depth of up to 10 cm. It is not sufficiently mobile to avoid substratum removal. Thus removal of the substratum would also remove the resident population of Brissopsis lyrifera and intolerance has been assessed to be high. However, Brissopsis lyrifera is likely to repopulate rapidly, see additional information below. | High | High | Moderate | High |
Smothering [Show more]SmotheringBenchmark. All of the population of a species or an area of a biotope is smothered by sediment to a depth of 5 cm above the substratum for one month. Impermeable materials, such as concrete, oil, or tar, are likely to have a greater effect. Further details. EvidenceBrissopsis lyrifera lives buried in muddy sediments up to 2-5 cm deep. As an urchin adapted for burrowing it is unlikely that it will be intolerant of additional sediment at the benchmark level and intolerance has been assessed to be low. However, it should be remembered that smothering by impermeable or viscous materials are likely to have some effect upon the animal, e.g. by causing deoxygenation. | Low | Immediate | Not sensitive | Moderate |
Increase in suspended sediment [Show more]Increase in suspended sedimentBenchmark. An arbitrary short-term, acute change in background suspended sediment concentration e.g., a change of 100 mg/l for one month. The resultant light attenuation effects are addressed under turbidity, and the effects of rapid settling out of suspended sediment are addressed under smothering. Further details EvidenceBrissopsis lyrifera is a non-selective deposit feeder and therefore does not rely on suspended food. However, for most benthic deposit feeders, food is suggested to be a limiting factor for body and gonad growth, at least between events of sedimentation of fresh organic matter (Hargrave, 1980; Tenore, 1988). Consequently, an increase in the suspended matter settling out from the water column to the substratum will be utilizable by Brissopsis lyrifera as a food resource. Although characteristically a sub-surface deposit feeder, Brissopsis lyrifera has been observed to increase its surface feeding (apical tuft becomes visible) activity after addition of organic matter to the sediment surface and utilized the material for growth (Hollertz et al., 1998; Hollertz, 1998). This suggests that an increase in siltation may be beneficial to the population and the species has been assesses as tolerant. | Tolerant* | Not relevant | Not sensitive* | Moderate |
Decrease in suspended sediment [Show more]Decrease in suspended sedimentBenchmark. An arbitrary short-term, acute change in background suspended sediment concentration e.g., a change of 100 mg/l for one month. The resultant light attenuation effects are addressed under turbidity, and the effects of rapid settling out of suspended sediment are addressed under smothering. Further details EvidenceBrissopsis lyrifera is a non-selective deposit feeder and therefore does not rely on suspended food. However, for most benthic deposit feeders, food is suggested to be a limiting factor for body and gonad growth, at least between events of sedimentation of fresh organic matter (Hargrave, 1980; Tenore, 1988). A decrease in siltation will increase competition for food resources and consequently impair growth. Intolerance has been assessed to be low. Recovery is likely to be rapid once food availability increases. | Low | Very high | Very Low | Moderate |
Desiccation [Show more]Desiccation
EvidencePopulations of Brissopsis lyrifera are subtidal and are not likely to be affected by desiccation. Where Brissopsis lyrifera occurs in coastal marine inlets and sealochs it is protected from desiccation because it inhabits a burrow in sediments to a depth of up to 10 cm. However, Brissopsis lyrifera is likely to be intolerant of continuous exposure to air and sunshine for one hour, e.g. as a result of by-catch and surface exposure. | Not relevant | Not relevant | Not relevant | Moderate |
Increase in emergence regime [Show more]Increase in emergence regimeBenchmark. A one hour change in the time covered or not covered by the sea for a period of one year. Further details EvidenceAn assessment of this factor is not relevant for Brissopsis lyrifera as the species lives subtidally. | Not relevant | Not relevant | Not relevant | Moderate |
Decrease in emergence regime [Show more]Decrease in emergence regimeBenchmark. A one hour change in the time covered or not covered by the sea for a period of one year. Further details EvidenceAn assessment of this factor is not relevant for Brissopsis lyrifera as the species lives subtidally. | Not relevant | Not relevant | Not relevant | Moderate |
Increase in water flow rate [Show more]Increase in water flow rateA change of two categories in water flow rate (view glossary) for 1 year, for example, from moderately strong (1-3 knots) to very weak (negligible). Further details EvidenceBrissopsis lyrifera is characteristic of offshore muddy sedimentary habitats exposed to only weak or very weak currents. The species is likely to be intolerant of the benchmark increase in water flow rate. Increased currents may wash out some of the sediments in which it burrows and decrease siltation of suspended organic matter to the sediment surface. The combination of a reduction in habitat and reduced food supply is likely to affect the viability of the population for a period of a year, so intolerance has been assessed to be intermediate. Brissopsis lyrifera is likely to recover rapidly, see additional information below. | Intermediate | High | Low | Low |
Decrease in water flow rate [Show more]Decrease in water flow rateA change of two categories in water flow rate (view glossary) for 1 year, for example, from moderately strong (1-3 knots) to very weak (negligible). Further details EvidenceBrissopsis lyrifera is characteristic of offshore and shallower, stable muddy habitats exposed to only weak or very weak currents. Sediments may become muddier due to increased settlement of silt if current strength declines. However, at the level of the benchmark it is not expected that populations will be affected. Thus Brissopsis lyrifera was assessed to be tolerant of a decrease in water flow rate. | Tolerant | Not relevant | Not sensitive | Low |
Increase in temperature [Show more]Increase in temperature
For intertidal species or communities, the range of temperatures includes the air temperature regime for that species or community. Further details EvidenceNo information specifically concerning the temperature tolerances of Brissopsis lyrifera was found. The geographic range of the species suggests that Brissopsis lyrifera is tolerant to a range of temperatures and a long term chronic increase of 2°C is unlikely to have adverse effects on populations around Britain and Ireland. Brissopsis lyrifera is an infaunal species that burrows up to 10 cm in depth, thus the burrow may offer some initial protection to an acute increase in water temperature. Furthermore, burrowing itself has been found to be significantly affected by temperature. Hollertz & Duchêne, (2001) found that Brissopsis lyrifera reworked almost double the amount of sediment per hour at 14 °C compared to activity at 7 °C (see adult general biology). However, Brissopsis lyrifera maintains a continuous contact with the overlying water column through the funnel (Hollertz, 2002). Increased water temperature may enhance microbial decomposition within the muddy sediments and promote deoxygenation to which Brissopsis lyrifera is intolerant. It is likely that Brissopsis lyrifera would be intolerant of an acute short term temperature increase and intolerance is assessed as intermediate. Brissopsis lyrifera has a high capacity for recovery, please see additional information below. | Intermediate | High | Low | Very low |
Decrease in temperature [Show more]Decrease in temperature
For intertidal species or communities, the range of temperatures includes the air temperature regime for that species or community. Further details EvidenceNo information specifically concerning the temperature tolerances of Brissopsis lyrifera was found. The geographic range of the species suggests that Brissopsis lyrifera is tolerant to a range of temperatures and a long term chronic decrease is unlikely to have adverse effects on populations around Britain and Ireland. Brissopsis lyrifera is an infaunal species that burrows up to 10 cm in depth, thus the burrow may offer some protection to acute decreases in temperature. Furthermore, Brissopsis lyrifera populations live in deep water so that events such as a very cold winter is unlikely to affect the temperature of these deeper waters. However, burrowing itself has been found to be significantly affected by temperature. Hollertz & Duchêne, (2001) found that Brissopsis lyrifera reworked almost half the amount of sediment per hour at 7 °C compared to activity at 14 °C (see adult general biology). Furthermore, Brissopsis lyrifera maintains a continuous contact with the overlying water column through the funnel (Hollertz, 2002). Very low water temperatures have caused mass mortalities of other similar echinoderms, such as Echinocardium cordatum. In the severe winter of 1962-63 masses of dead Echinocardium cordatum were observed in regions of the North Sea and English Channel (Crisp (ed.), 1964), although it was reported that living specimens were obtained easily enough by digging (Moyse & Nelson-Smith, 1964 in Crisp (ed.), 1964). Intolerance of Brissopsis lyrifera to acute decreases in temperature has been assessed to be intermediate. Brissopsis lyrifera has a high capacity for recovery, see additional information below. | Intermediate | High | Low | |
Increase in turbidity [Show more]Increase in turbidity
EvidenceBrissopsis lyrifera is likely to have a poor facility for perception of irradiance and consequently is likely to be tolerant of an increase in turbidity. However, an increase in turbidity may be indicative of a potential increase in the supply of organic matter that may settle upon the sediment surface and be utilized as a food resource by Brissopsis lyrifera (Hollertz, 1998). | Tolerant* | Not relevant | Not sensitive* | Moderate |
Decrease in turbidity [Show more]Decrease in turbidity
EvidenceBrissopsis lyrifera is likely to have a poor facility for perception of irradiance and consequently is probably tolerant of a decrease in turbidity. A decrease in turbidity may be indicative of a reduced supply of organic matter eventually falling to the sediment surface which the species can utilize as a food resource (see changes in siltation). Reduced growth and fecundity may result. | Low | Very high | Very Low | Moderate |
Increase in wave exposure [Show more]Increase in wave exposureA change of two ranks on the wave exposure scale (view glossary) e.g., from Exposed to Extremely exposed for a period of one year. Further details EvidenceBrissopsis lyrifera is characteristic of offshore and stable muddy nearshore habitats, where wave exposure is negligible. Populations of Brissopsis lyrifera occurring offshore at depths greater than 60 m would not be subjected to exposure from wave action, as the effects are attenuated with increasing depth. Populations situated in normally sheltered stable habitats at shallower depths may experience some disturbance to the sediment surface. However, as Brissopsis lyrifera burrows in the sediment to a depth of 10 cm, it is unlikely that turnover and displacement would occur to an extent where the population is significantly reduced. Intolerance has been assessed to be low. | Low | High | Low | Low |
Decrease in wave exposure [Show more]Decrease in wave exposureA change of two ranks on the wave exposure scale (view glossary) e.g., from Exposed to Extremely exposed for a period of one year. Further details EvidenceBrissopsis lyrifera is characteristic of offshore and stable muddy nearshore habitats where wave exposure is negligible. Thus an assessment for decreased wave exposure was not considered relevant. | Not relevant | Not relevant | Not relevant | Low |
Noise [Show more]Noise
EvidenceNo information concerning sound or vibration reception in echinoids was found. | No information | Not relevant | No information | Not relevant |
Visual presence [Show more]Visual presenceBenchmark. The continuous presence for one month of moving objects not naturally found in the marine environment (e.g., boats, machinery, and humans) within the visual envelope of the species or community under consideration. Further details EvidenceSome response to visual disturbance has been detected in echinoderms. However, Brissopsis lyrifera lives buried in muddy substrates up to 10 cm deep thus visual disturbance was not considered relevant to this species. | No information | Not relevant | No information | Not relevant |
Abrasion & physical disturbance [Show more]Abrasion & physical disturbanceBenchmark. Force equivalent to a standard scallop dredge landing on or being dragged across the organism. A single event is assumed for assessment. This factor includes mechanical interference, crushing, physical blows against, or rubbing and erosion of the organism or habitat of interest. Where trampling is relevant, the evidence and trampling intensity will be reported in the rationale. Further details. EvidenceBrissopsis lyrifera has a fragile test that is likely to be damaged by an abrasive force, such as movement of trawling gear. In areas of the North Sea where heavy demersal fishing for Nephrops norvegicus occurs, populations of Brissopsis lyrifera may be reduced owing to damage inflicted to the 'test' by the fishing gear. Broken tests may be seen on the seabed (E.I.S. Rees, M. Costello, pers comm. to Connor et al., 1997). Intolerance has been assessed to be intermediate as some individuals of the species may be destroyed by the abrasive factor. Populations of Brissopsis lyrifera are likely to recover from effects of physical disturbance rapidly as the species is fecund and recruits annually with pelagic larva. | Intermediate | High | Low | High |
Displacement [Show more]DisplacementBenchmark. Removal of the organism from the substratum and displacement from its original position onto a suitable substratum. A single event is assumed for assessment. Further details EvidenceIf displaced to the substratum surface Brissopsis lyrifera is potentially exposed to an increased risk of predation, but is capable of re-burrowing into the sediment. In the laboratory, animals left on the sediment surface were all observed to have buried themselves within a few hours (Hollertz, 2002). Intolerance is assessed to be low. Echinocardium cordatum can re-burrow into the sediment within 20 minutes, and as a similar specialized burrower it is also likely that Brissopsis lyrifera will submerge as rapidly, thus recovery has been assessed to be immediate. | Low | Immediate | Not sensitive | Low |
Chemical pressures
Use [show more] / [show less] to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Synthetic compound contamination [Show more]Synthetic compound contaminationSensitivity is assessed against the available evidence for the effects of contaminants on the species (or closely related species at low confidence) or community of interest. For example:
The evidence used is stated in the rationale. Where the assessment can be based on a known activity then this is stated. The tolerance to contaminants of species of interest will be included in the rationale when available; together with relevant supporting material. Further details. EvidenceDahllöf et al., (1999) studied the long term effects of tri-n-butyl-tin (TBT) on the function of a marine sediment system. TBT spiked sediment was added to a sediment that already had a TBT background level of approximately 27 ng g-1 and contained the following fauna: Amphiura spp., Brissopsis lyrifera and several species of polychaete. Within two days of treatment with a TBT concentration above 13.7 µmol /m² all species except the polychaetes had crept up to the surface and after six weeks these fauna had started to decay. Thus contamination from TBT is likely to result cause death of this species. Detergents used to disperse oil from the Torrey Canyon oil spill caused mass mortalities of a similar species, Echinocardium cordatum (Smith, 1968). Sea-urchins, especially the eggs and larvae are used for toxicity testing and environmental monitoring (reviewed by Dinnel et al., 1998). It is likely therefore, that Brissopsis lyrifera and its larvae are highly intolerant of organic compounds. Brissopsis lyrifera populations would probably recover rapidly assuming deterioration of contaminants, see additional information below. | High | High | Moderate | Low |
Heavy metal contamination [Show more]Heavy metal contaminationEvidenceInformation concerning the effects of heavy metals on echinoderms is limited and no information specific to Brissopsis lyrifera was found. | No information | No information | No information | Not relevant |
Hydrocarbon contamination [Show more]Hydrocarbon contaminationEvidenceNo information directly concerning the intolerance of Brissopsis lyrifera to hydrocarbons was found. Suchanek (1993), proposed that echinoderms would be especially sensitive to the toxic effects of oil, owing to the exposure of the epidermis. However, as Brissopsis lyrifera burrows in muddy sediments to a depth of up to 10 cm, the likely route of exposure to the animal will arise from its feeding method. As a non-selective deposit feeder Brissopsis lyrifera is likely to ingest contaminated sediments. Intolerance has been assessed to be intermediate owing to a range of effects reported in other echinoderms (mortalities, feeding and growth inhibition, embryological abnormalities) reviewed by Suchanek (1993). Brissopsis lyrifera is likely to recover from this factor, see additional information below. | Intermediate | High | Low | Moderate |
Radionuclide contamination [Show more]Radionuclide contaminationEvidenceInsufficient | No information | Not relevant | No information | Not relevant |
Changes in nutrient levels [Show more]Changes in nutrient levelsEvidenceBrissopsis lyrifera is characteristic of muddy sediments with significant organic matter content and is a non-selective deposit feeder. For most benthic deposit feeders, food is suggested to be a limiting factor for body and gonad growth, at least between events of sedimentation of fresh organic matter (Hargrave, 1980; Tenore, 1988). An increase in nutrient availability is likely to promote pelagic productivity and increase the amount of organic matter reaching the sea bed. Hollertz (1998) demonstrated that such organic matter is utilizable by Brissopsis lyrifera as a food resource for gonad growth. Thus Brissopsis lyrifera may benefit from some nutrient enrichment. However, an excess of nutrients (eutrophication) facilitating a high pelagic production, in combination with thermal stratification of the water column in summer is likely to cause mortality of Brissopsis lyrifera indirectly, owing to the effects of hypoxia. Refer to 'changes in oxygenation' for a separate assessment of intolerance. | Tolerant* | Not relevant | Not sensitive* | High |
Increase in salinity [Show more]Increase in salinity
EvidenceEchinoderms are stenohaline owing to the lack of an excretory organ and a poor ability to osmo- and ion-regulate (Stickle & Diehl, 1987). The inability of echinoderms to osmoregulate extracellularly causes body fluid volume to increase or decrease when individuals are exposed to higher salinity. No information concerning the specific tolerance of Brissopsis lyrifera to changes in salinity was found. However, the preferred habitat of Brissopsis lyrifera is found in waters of full salinity where an increase of salinity is not likely to occur. | Not relevant | Not relevant | Not relevant | Low |
Decrease in salinity [Show more]Decrease in salinity
EvidenceEchinoderms are stenohaline owing to the lack of an excretory organ and a poor ability to osmo- and ion-regulate (Stickle & Diehl, 1987). The inability of echinoderms to osmoregulate extracellularly causes body fluid volume to or decrease when individuals are exposed to lower salinities. However, no information concerning the specific tolerance of Brissopsis lyrifera to a decrease in salinity was found. Populations that occur in sheltered nearshore situations, such as sealochs, which periodically receive inputs of freshwater are unlikely to experience the reduced salinities recorded at the surface. Furthermore, the muddy sediment in which Brissopsis lyrifera burrows may offer some protection. However, as a species with an apparent preference for fully saline conditions it is likely to be intolerant of a decrease in salinity. | Low | High | Low | |
Changes in oxygenation [Show more]Changes in oxygenationBenchmark. Exposure to a dissolved oxygen concentration of 2 mg/l for one week. Further details. EvidenceBrissopsis lyrifera is reported as a species sensitive to hypoxia (Diaz & Rosenberg, 1995). Demersal trawling in hypoxic areas has shown that large numbers of infaunal species in areas of high abundance and biomass leave their positions in the sediment and lie exposed on the bottom. At a bottom water oxygen concentration of ca. 1 ml/l (15% saturation) in the Kattegat, Baden et al., (1990) caught no fishes, but 200-400 kg per hour of benthic invertebrates that included the echinoderms Brissopsis lyrifera and Echinocardium cordatum. Similar mass migration of benthic infauna (including Brissopsis lyrifera) to the sediment surface was recorded during trawling in the North Sea with low values of oxygen (ca 2 ml/l) recorded (Dyer et al., 1983). Hollertz (2002) reported that Brissopsis lyrifera could tolerate ca 4 ml/l (ca 5.6 mg/l) for at least 15 hours in the laboratory and that the animals recovered quickly. However, this is a much higher oxygen concentration than that in the benchmark. Recovery of Brissopsis lyrifera has been assessed to be moderate. Recovery of 'mature' equilibrium dominants eliminated by hypoxia appears to be a process requiring more than two years (Diaz & Rosenberg, 1995). In the Gullmarsfjord (where Brissopsis lyrifera is reported; Brattström, 1946; Vasseur & Carlsen, 1949), the 1980/1981 hypoxia, ca 0.2 ml/l, eliminated all the macrobenthic fauna below 115 m depth. The recovery sequence was slow and communities were not re-established eighteen months after the collapse (Josefson & Widbom, 1988). | High | Moderate | Moderate | High |
Biological pressures
Use [show more] / [show less] to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Introduction of microbial pathogens/parasites [Show more]Introduction of microbial pathogens/parasitesBenchmark. Sensitivity can only be assessed relative to a known, named disease, likely to cause partial loss of a species population or community. Further details. EvidenceThe occurrence of the ascothoracidan parasite Ulophysema öresundense (Brattström) has been observed in the body cavity of Brissopsis lyrifera (Brattström, 1946). This parasite may cause sexual castration but no further information concerning the effect of this parasite on the population was found. | No information | Not relevant | No information | Low |
Introduction of non-native species [Show more]Introduction of non-native speciesSensitivity assessed against the likely effect of the introduction of alien or non-native species in Britain or Ireland. Further details. EvidenceNo alien or non-native species are known to compete with Brissopsis lyrifera. | No information | Not relevant | No information | Not relevant |
Extraction of this species [Show more]Extraction of this speciesBenchmark. Extraction removes 50% of the species or community from the area under consideration. Sensitivity will be assessed as 'intermediate'. The habitat remains intact or recovers rapidly. Any effects of the extraction process on the habitat itself are addressed under other factors, e.g. displacement, abrasion and physical disturbance, and substratum loss. Further details. EvidenceBrissopsis lyrifera is not a targeted species for harvest, but it may be affected by the extraction of other species, please refer to 'Extraction of other species'. | Not relevant | Not relevant | Not relevant | Low |
Extraction of other species [Show more]Extraction of other speciesBenchmark. A species that is a required host or prey for the species under consideration (and assuming that no alternative host exists) or a keystone species in a biotope is removed. Any effects of the extraction process on the habitat itself are addressed under other factors, e.g. displacement, abrasion and physical disturbance, and substratum loss. Further details. EvidenceMud in deep offshore waters may be characterized by urchin Brissopsis lyrifera and brittle star Amphiura chiajei, communities. In certain sea areas around the British Isles, e.g. northern Irish Sea, this community may also contain the Norway lobster, Nephrops norvegicus. Nephrops norvegicus is one of the most important fisheries in Scotland and the community can consequently become the focus for fishing activity (Mackie, Oliver & Rees, 1995). Where heavy demersal fishing occurs populations of Brissopsis lyrifera may be reduced owing to damage inflicted to the 'test' by the fishing gear. Broken tests may be seen on the sea bed (E.I.S. Rees, M. Costello pers. Comm. In Connor et al., 1997). The impact of pots/creels on the sea bed and non-target species is considered to be slight. Brissopsis lyrifera is likely to recover from fishing impacts as the species is fecund and recruits annually with pelagic larva. | Intermediate | High | Low | Moderate |
Additional information
Recoverability. Brissopsis lyrifera is likely to have a high capacity for recovery. The species is fecund and has shown clear evidence of successful and consecutive annual recruitment (Buchanan, 1967). The adults are burrowers, so the larval phase is the main dispersive mechanism of the urchin. Echinoderm larvae undergo a complicated and protracted metamorphosis in the plankton. For instance, the larvae of other echinoderms, %Echinocardium cordatum% and %Echinus esculentus% remain in the plankton for 40 and 46-60 days respectively (Kashenko, 1994; MacBride, 1914). Thus the larvae of Brissopsis lyrifera probably remain in the plankton for sufficient time to be swept away from their spawning ground to new areas or to re-populate disturbed areas. Whilst, recolonization is likely to occur rapidly, the new population will not reach reproductive maturity for about four years (Buchanan, 1967).
Importance review
Policy/legislation
- no data -
Status
| National (GB) importance | - | Global red list (IUCN) category | - |
Non-native
| Parameter | Data |
|---|---|
| Native | - |
| Origin | - |
| Date Arrived | - |
Importance information
Brissopsis lyrifera burrows down to ca. 2-5 cm in the sediment, where it ingests sediment in bulk and feeds on the associated organic matter. Brissopsis lyrifera is therefore an active 'bioturbator'. Bioturbation is particularly important in controlling chemical, physical and biological processes in marine sediments, especially when the influences of physical disturbances such as wave action or strong currents are minimized (Widdicombe & Austen, 1999).
Brissopsis lyrifera plays a role in the enhancement of regional species heterogeneity in an otherwise largely homogenous environment (Widdicombe et al., 2000; Widdicombe & Austen, 1999, 1998; Austen, Widdicombe & Villano-Pitacco, 1998; Austen & Widdicombe, 1998). When burrowing, Brissopsis lyrifera disturbs the sediment in a way that may result in lowered sediment stability (De Ridder & Lawrence, 1982). This disturbance combined with its respiratory activity alters the sediment chemistry, probably increasing the oxygenation of the sediment at deeper levels. Consequently, the effects of Brissopsis lyrifera on the associated meiofauna will arise through both its non-selective feeding habit and its alteration of the physical and chemical environment of the sediment in which the meiofauna live. Widdicombe & Austen (1998) found that the presence of Brissopsis lyrifera had significant effects on the fluxes of two nutrients. Burrowing promoted oxygen penetration into the sediment and led to a decrease in nitrate reduction, suggesting that bioturbating macrofauna, such as Brissopsis lyrifera, can regulate denitrification.
Bibliography
Baden, S.P., Loo, L.O., Pihl, L. & Rosenberg, R., 1990. Effects of eutrophication on the benthic communities including fish - Swedish west coast. Ambio, 19, 113-122.
Brattström, H., 1946. Observations on Brissopsis lyrifera (Forbes) in the Gullmar Fjord. Arkive fur Zoologie, 37A, 1-27.
Bruce, J.R., Colman, J.S. & Jones, N.S., 1963. Marine fauna of the Isle of Man. Liverpool: Liverpool University Press.
Bryan, G.W., 1984. Pollution due to heavy metals and their compounds. In Marine Ecology: A Comprehensive, Integrated Treatise on Life in the Oceans and Coastal Waters, vol. 5. Ocean Management, part 3, (ed. O. Kinne), pp.1289-1431. New York: John Wiley & Sons.
Buchanan, J.B., 1967. Dispersion and demography of some infaunal echinoderm populations. Symposia of the Zoological Society of London, 20, 1-11.
Connor, D.W., Dalkin, M.J., Hill, T.O., Holt, R.H.F. & Sanderson, W.G., 1997a. Marine biotope classification for Britain and Ireland. Vol. 2. Sublittoral biotopes. Joint Nature Conservation Committee, Peterborough, JNCC Report no. 230, Version 97.06., Joint Nature Conservation Committee, Peterborough, JNCC Report no. 230, Version 97.06.
Crisp, D.J. (ed.), 1964. The effects of the severe winter of 1962-63 on marine life in Britain. Journal of Animal Ecology, 33, 165-210.
Crothers, J.H. (ed.), 1966. Dale Fort Marine Fauna. London: Field Studies Council.
Dahllöf, I., Blanck, H., Hall, P.O.J. & Molander, S., 1999. Long term effects of tri-n-butyl-tin on the function of a marine sediment system. Marine Ecology Progress Series, 188, 1-11.
De Ridder, C. & Lawrence, J.M., 1982. Food and feeding mechanisms: Echinoidea. In Echinoderm Nutrition (ed. Jangoux, M. & Lawrence, J.M.), pp. 57-115. Rotterdam: A.A. Balkema.
Diaz, R.J. & Rosenberg, R., 1995. Marine benthic hypoxia: a review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanography and Marine Biology: an Annual Review, 33, 245-303.
Dinnel, P.A., Pagano, G.G., & Oshido, P.S., 1988. A sea urchin test system for marine environmental monitoring. In Echinoderm Biology. Proceedings of the Sixth International Echinoderm Conference, Victoria, 23-28 August 1987, (R.D. Burke, P.V. Mladenov, P. Lambert, Parsley, R.L. ed.), pp 611-619. Rotterdam: A.A. Balkema.
Dyer, M.F., Pope, J.G., Fry. P.D., Law, R.J. & Portmann, J.E., 1983. Changes in fish and benthos catches off the Danish coast in September 1981. Journal of the Marine Biological Association of the United Kingdom, 63, 767-775.
Ferrand, J.G., Vadon, C., Doumenc, D & Guille, A., 1988. The effect of depth on the reproductive cycle of Brissopsis lyrifera (Echinoidea, Echinodermata) in the Gulf of Lions, Mediterranean Sea. Marine Biology, 99, 387-392.
Fish, J.D. & Fish, S., 1996. A student's guide to the seashore. Cambridge: Cambridge University Press.
Foster-Smith, J. (ed.), 2000. The marine fauna and flora of the Cullercoats District. Marine species records for the North East Coast of England. Sunderland: Penshaw Press, for the Dove Marine Laboratory, University of Newcastle upon Tyne.
Hargrave, B.T., 1980. Factors affecting the flux of organic matter to sediments in a marine bay. In Marine Benthic Dynamics (eds. Tenore, K.R. & Coull, B.C.), 243-263. USA: University of South Carolina Press.
Hayward, P., Nelson-Smith, T. & Shields, C. 1996. Collins pocket guide. Sea shore of Britain and northern Europe. London: HarperCollins.
Hayward, P.J. & Ryland, J.S. (ed.) 1995b. Handbook of the marine fauna of North-West Europe. Oxford: Oxford University Press.
Hollertz, K. & Duchêne, J.-C., 2001. Burrowing behaviour and sediment reworking in the heart urchin Brissopsis lyrifera Forbes (Spatangoida). Marine Biology, 139, 951-957.
Hollertz, K., 1998. The response of Brissopsis lyrifera (Echinoidea: Spatangoida) to organic matter on the sediment surface. In Echinoderm Research (eds. Candia Carnevali, M.D. & Bonasoro, F.), 79-84.
Hollertz, K., 2002. Feeding biology and carbon budget of the sediment-burrowing heart urchin Brissopsis lyrifera (Echinoidea: Spatangoida). Marine Biology, 140, 959-969.
Hollertz, K., Skold, M. & Rosenberg, R., 1998. Interactions between two deposit feeding echinoderms: the spatangoid Brissopsis lyrifera (Forbes) and the ophiuroid Amphiura chiajei (Forbes). Hydrobiologia, 376, 287-295.
Howson, C.M. & Picton, B.E., 1997. The species directory of the marine fauna and flora of the British Isles and surrounding seas. Belfast: Ulster Museum. [Ulster Museum publication, no. 276.]
JNCC (Joint Nature Conservation Committee), 1999. Marine Environment Resource Mapping And Information Database (MERMAID): Marine Nature Conservation Review Survey Database. [on-line] http://www.jncc.gov.uk/mermaid
Kashenko, S.D., 1994. Larval development of the heart urchin Echinocardium cordatum feeding on different macroalgae. Biologiya Morya, 20, 385-389.
Kinne, O. (ed.), 1984. Marine Ecology: A Comprehensive, Integrated Treatise on Life in Oceans and Coastal Waters.Vol. V. Ocean Management Part 3: Pollution and Protection of the Seas - Radioactive Materials, Heavy Metals and Oil. Chichester: John Wiley & Sons.
Lawrence, J.M., 1996. Mass mortality of echinoderms from abiotic factors. In Echinoderm Studies Vol. 5 (ed. M. Jangoux & J.M. Lawrence), pp. 103-137. Rotterdam: A.A. Balkema.
MacBride, E.W., 1914. Textbook of Embryology, Vol. I, Invertebrata. London: MacMillan & Co.
Mortensen, T.H., 1927. Handbook of the echinoderms of the British Isles. London: Humphrey Milford, Oxford University Press.
Moyse, J. & Nelson-Smith, A., 1964. Effects of the severe cold of 1962-63 upon shore animals in South Wales. Journal of Animal Ecology, 33, 183-190.
Nichols, D., 1969. Echinoderms (4th ed.). London: Hutchinson & Co.
Picton, B.E. & Costello, M.J., 1998. BioMar biotope viewer: a guide to marine habitats, fauna and flora of Britain and Ireland. [CD-ROM] Environmental Sciences Unit, Trinity College, Dublin.
Picton, B.E., 1993. A field guide to the shallow-water echinoderms of the British Isles. London: Immel Publishing Ltd.
Rumohr, H. & Kujawski, T., 2000. The impact of trawl fishery on the epifauna of the southern North Sea. ICES Journal of Marine Science, 57, 1389-1394.
Schinner, G.O., 1993. Burrowing behaviour, substratum preference and distribution of Schisater canaliferus (Echinoidea: Spatangoida) in the Northern Adriatic Sea. Marine Ecology, 14, 129-145.
Stronkhorst, J., Hattum van, B. & Bowmer, T., 1999. Bioaccumulation and toxicity of tributyltin to a burrowing heart urchin and an amphipod in spiked, silty marine sediments. Environmental Toxicology and Chemistry, 18 (10), 2343-2351. DOI https://doi.org/10.1002/etc.5620181031
Suchanek, T.H., 1993. Oil impacts on marine invertebrate populations and communities. American Zoologist, 33, 510-523. DOI https://doi.org/10.1093/icb/33.6.510
Tenore, K.R., 1988. Nitrogen in benthic food chains. In Nitrogen Cycling in Coastal Marine Environments, (eds. Blackburn, T.H. & Sörensen J.), 191-206. New York: John Wiley & Sons Ltd.
Tunberg, B., 1991. Övervakning av mjukbottenfauna längs Sveriges västkust. Swedish Environmental Protection Network 4066.
Ursin, E., 1960. A quantitative investigation of the echinoderm fauna of the central North Sea. Meddelelser fra Danmark Fiskeri-og-Havundersogelser, 2 (24), pp. 204.
Vasseur, E. & Carlsen, I., 1949. Sexual maturity of the sea urchin, Brissopsis lyrifera (Forbes) in the Gullmar Fjord. Arkive fur Zoologie, 16, 1-10.
Widdicombe, S. & Austen, M.C., 1998. Experimental evidence for the role of Brissopsis lyrifera (Forbes, 1841) as a critical species in the maintenance of benthic diversity and the modification of sediment chemistry. Journal of Experimental Marine Biology and Ecology, 228, 241-255.
Widdicombe, S. & Austen, M.C., 1999. Mesocosm investigation into the effects of bioturbation on the diversity and structure of a subtidal macrobenthic community. Marine Ecology Progress Series, 189, 181-193.
Widdicombe, S., Austen, M.C., Kendall, M.A., Warwick, R.M., & Jones, M.B., 2000. Bioturbation as a mechanism for setting and maintaining levels of diversity in subtidal macrobenthic communities. Hydrobiologia, 440, 369-377.
Datasets
NBN (National Biodiversity Network) Atlas. Available from: https://www.nbnatlas.org.
OBIS (Ocean Biodiversity Information System), 2024. Global map of species distribution using gridded data. Available from: Ocean Biogeographic Information System. www.iobis.org. Accessed: 2024-04-17
Citation
This review can be cited as:
Last Updated: 29/10/2004