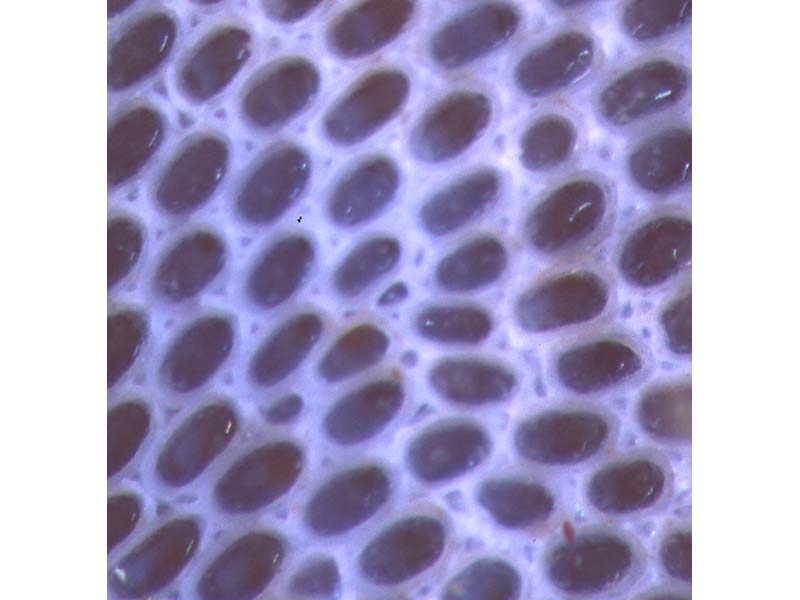
An encrusting bryozoan (Conopeum reticulum)
Distribution data supplied by the Ocean Biodiversity Information System (OBIS). To interrogate UK data visit the NBN Atlas.Map Help
| Researched by | Dr Harvey Tyler-Walters & Susie Ballerstedt | Refereed by | Dr Peter J. Hayward |
| Authority | (Linnaeus, 1767) | ||
| Other common names | - | Synonyms | - |
Summary
Description
Conopeum reticulum colonies form extensive gauze-like encrustations. Individuals (zooids) within the colony are approximately 0.4-0.6 x 0.2-0.3 mm in size, elongate, rectangular or polygonal in outline with a thickened, finely granular margin. The margin occasionally bears a few thin, pointed, delicate spines. The upper (frontal) surface of zooids is membranous with a semicircular light-brown operculum at one end. Triangular dwarf, non-feeding zooids are often present at the distal end of zooids in the gaps between the normal zooids.
Recorded distribution in Britain and Ireland
Recorded in a few locations on the west coast of Scotland (but probably more widespread (P. Hayward, pers. comm.)) and the north east of England. It is more common around the coast of Wales and along the south coast of Britain.Global distribution
Present in the Kattegat and shores of the southern North Sea. Recently recorded in the western Mediterranean.Habitat
Conopeum reticulum favours estuaries and bays on sheltered coasts. Found from the lower shore into the shallow sublittoral, on hard substrata, shells, stones, and submerged structures. Not usually associated with plants. In brackish areas, it is often abundant on the inner sides of oyster valves (Ostrea edulis). Rarely found off-shore.Depth range
Intertidal to at least 42mIdentifying features
- Forms extensive gauze-like encrustations.
- Individuals (zooids) oval, approximately 0.4-0.6 x 0.2-0.3 mm in size.
- Triangular dwarf non-feeding heterozooids present between normal zooids, occasionally forming a dumbbell shape at the distal edge of the zooid.
- Operculum is broad, semicircular, light brown with a folded membranous edge.
- Polypide with 11 tentacles.
- Avicularia absent.
Additional information
Normal autozooids occasionally become irregularly shaped or larger, especially at the edges of colonies where there are sometimes large, irregular gaps to fill.
Listed by
- none -
Biology review
Taxonomy
| Level | Scientific name | Common name |
|---|---|---|
| Phylum | Bryozoa | Sea mats, horn wrack & lace corals |
| Class | Gymnolaemata | Naked throat bryozoans |
| Order | Cheilostomatida | |
| Family | Electridae | |
| Genus | Conopeum | |
| Authority | (Linnaeus, 1767) | |
| Recent Synonyms | ||
Biology
| Parameter | Data | ||
|---|---|---|---|
| Typical abundance | Low density | ||
| Male size range | |||
| Male size at maturity | |||
| Female size range | Small-medium (3-10 cm) | ||
| Female size at maturity | |||
| Growth form | Crustose hard | ||
| Growth rate | See additional information | ||
| Body flexibility | None (less than 10 degrees) | ||
| Mobility | Sessile, permanent attachment | ||
| Characteristic feeding method | Active suspension feeder | ||
| Diet/food source | Planktotroph | ||
| Typically feeds on | Phytoplankton (<50µm), macroalgal spores, detritus, and bacteria. | ||
| Sociability | Colonial | ||
| Environmental position | Epifaunal | ||
| Dependency | Independent. | ||
| Supports | None | ||
| Is the species harmful? | No | ||
Biology information
Growth rates. Growth, measured in zooid number, is exponential in Conopeum reticulum (Menon, 1972). Growth rates in bryozoans have been shown to vary with environmental conditions, especially, food supply, temperature, competition for food and space, and genotype. For example, although growth rates increased with temperature, zooid size decreased, which may be due to increased metabolic costs at higher temperature (Menon, 1972; Ryland, 1976; Hunter & Hughes, 1994). Menon (1972) reported that in culture, growth in Conopeum reticulum reached a plateau after about 30 days and that the growth rate had significantly reduced at the end of 6 months. In his experiments, Conopeum reticulum colonies grew to ca 1000 zooids within ca 28 days at 12°C and ca 18 days at 22°C, although these rates were slower than under natural conditions (Menon, 1972). Feeding rates also varied with respect to temperature (Menon, 1974).
Feeding. The structure and function of the bryozoan lophophore were reviewed by Ryland (1976), Winston (1977) and Hayward & Ryland (1998). Ambient water flow is important for bringing food-bearing water within the range of the colonies' own pumping ability (McKinney, 1986). Best & Thorpe (1994) suggested that intertidal Bryozoa would probably be able to feed on small flagellates, bacteria, algal spores and small pieces of abraded macroalgae.
Habitat preferences
| Parameter | Data |
|---|---|
| Physiographic preferences | Open coast, Strait or Sound, Ria or Voe, Estuary, Enclosed coast or Embayment |
| Biological zone preferences | Lower eulittoral, Lower infralittoral, Sublittoral fringe, Upper infralittoral |
| Substratum / habitat preferences | Macroalgae, Artificial (man-made), Bedrock, Caves, Cobbles, Large to very large boulders, Other species (see additional information), Overhangs, Pebbles, Rockpools, Small boulders, Under boulders |
| Tidal strength preferences | Moderately strong 1 to 3 knots (0.5-1.5 m/sec.), Strong 3 to 6 knots (1.5-3 m/sec.), Weak < 1 knot (<0.5 m/sec.) |
| Wave exposure preferences | Exposed, Extremely sheltered, Moderately exposed, Sheltered, Very sheltered |
| Salinity preferences | Full (30-40 psu), Reduced (18-30 psu), Variable (18-40 psu) |
| Depth range | Intertidal to at least 42m |
| Other preferences | No text entered |
| Migration Pattern | Non-migratory or resident |
Habitat Information
The distribution may be imprecise since Conopeum reticulum is frequently confused with other species but it is probably common off all British coasts (Hayward & Ryland, 1998). Conopeum reticulum has been reported from a wide variety of hard substrata including boulders, cobbles, shell and small stones on sediment, as well as the shells of Mytilus edulis. Colonies inhabiting stones and cobbles are probably ephemeral, removed by abrasion and rolling during winter storms. Grant & Hayward (1985) reported Conopeum reticulum in shallow water bryozoan assemblages in the English Channel at a mean depth of 42m.Life history
Adult characteristics
| Parameter | Data |
|---|---|
| Reproductive type | Budding |
| Reproductive frequency | Annual episodic |
| Fecundity (number of eggs) | See additional information |
| Generation time | <1 year |
| Age at maturity | Less than 1 year |
| Season | June - October |
| Life span | Insufficient information |
Larval characteristics
| Parameter | Data |
|---|---|
| Larval/propagule type | - |
| Larval/juvenile development | Planktotrophic |
| Duration of larval stage | 1-6 months |
| Larval dispersal potential | Greater than 10 km |
| Larval settlement period |
Life history information
Reproduction. Bryozoan colonies are hermaphrodite, however, zooids may be monoecious, dioecious, protandrous or protogynous, depending on the species (Hayward & Ryland, 1998). In most bryozoans the zooids are hermaphrodite and probably protandric, becoming male then female (Reed, 1991; Hayward & Ryland, 1998). Sperm are shed from pores in the polypide tentacles of male zooids (Hayward & Ryland, 1998). In bryozoans, released sperm are entrained by the tentacles of female polypides and may not disperse far, resulting in self-fertilization. However, genetic cross-fertilization is assumed in most bryozoans, although there is evidence of self-fertilization (Reed, 1991; Hayward & Ryland, 1998). Female zooids develop a ciliated intertentacular organ, which collects eggs from the ovaries, passes them to the gonopore, and expels them beyond the lophophores during spawning (Reed, 1991). Fertilization is thought to occur either within the tentacular organ or just as eggs are spawned (Ryland, 1976; Reed 1991). Conopeum reticulum breeds between June and early October in Britain and Ireland, and yellowish-white, rounded eggs (average size 110 by 80µm) were present from July to September in the River Crouch (Cook, 1964; Hayward & Ryland, 1998). Cook (1964) reported that eggs were rarely spawned in daylight but that many were found in the morning. Day length is an important cue for spawning in some coastal species of bryozoa that spawn in the first few hours of daylight (Hayward & Ryland, 1998). Cook (1964) reported that the intertentacular organ contained 5-9 eggs per zooid. However, while each individual zooid is not prolific, the fecundity of the colony is probably directly proportional to the number of functional zooids (Bayer et al., 1994) and is probably high. Although Conopeum reticulum colonies could probably survive for several years, it is probably adapted to ephemeral habitats, capable of rapid growth and reproduction of numerous offspring (r-selected). Larvae were present in the plankton in the same period (July to September) in the River Crouch and River Blackwater (Cook, 1964). Reed (1991) reported that planktotrophic cyphonautes larvae spend between one to three months in the plankton.
Recruitment. Bryozoan larvae are probably sensitive to surface contour, chemistry and the proximity of conspecific colonies. However, Hayward & Ryland (1998) suggested that larval behaviour at settlement is only of prime importance to species occupying ephemeral habitats. Eggleston (1972b) demonstrated that the number and abundance of species of bryozoan increased with increased current strength, primarily due to a resultant increase in the availability of stable, hard substrata (Eggleston, 1972b; Ryland, 1976). Ryland (1976) reported that significant settlement in bryozoans was only found near a reservoir of breeding colonies. Ryland (1977) suggested that marine bryozoan larvae tend to settle on the underside of submerged structures or in shaded habitats, possibly due to avoidance of accumulated sediment or competition from algae. However, Conopeum reticulum larvae have an extended planktonic life and Conopeum reticulum is a member of fouling communities (Ryland, 1967). In addition, Conopeum sp. has been reported to have spread into the Caspian Sea after the opening of the Volga-Don canal, possibly on shipping (Ryland 1967). Therefore, Conopeum reticulum probably exhibits good dispersal and potentially very rapid recruitment. For example, Hatcher (1998) reported that spring recruitment to an artificial reef in Poole Bay was dominated by tubeworms and encrusting bryozoans including Conopeum reticulum. Conopeum reticulum colonized artificial reef surfaces within 6 months from May to October 1991 (Hatcher, 1998).
Sensitivity review
The MarLIN sensitivity assessment approach used below has been superseded by the MarESA (Marine Evidence-based Sensitivity Assessment) approach (see menu). The MarLIN approach was used for assessments from 1999-2010. The MarESA approach reflects the recent conservation imperatives and terminology and is used for sensitivity assessments from 2014 onwards.
Physical pressures
Use / to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Substratum loss [Show more]Substratum lossBenchmark. All of the substratum occupied by the species or biotope under consideration is removed. A single event is assumed for sensitivity assessment. Once the activity or event has stopped (or between regular events) suitable substratum remains or is deposited. Species or community recovery assumes that the substratum within the habitat preferences of the original species or community is present. Further details EvidenceRemoval of the substratum, be it shell, rock, or cobble will result in removal of the attached colonies of Conopeum reticulum. Therefore, an intolerance of high has been recorded. Recoverability is likely to be very high (see additional information below). | High | Very high | Low | Low |
Smothering [Show more]SmotheringBenchmark. All of the population of a species or an area of a biotope is smothered by sediment to a depth of 5 cm above the substratum for one month. Impermeable materials, such as concrete, oil, or tar, are likely to have a greater effect. Further details. EvidenceSmothering by 5 cm of sediment is likely to prevent feeding, and hence growth and reproduction, as well as respiration. In addition, associated sediment abrasion may remove the bryozoan colonies. A layer of sediment will probably also interfere with larval settlement. Therefore, an intolerance of high has been recorded. Recoverability has been assessed as very high (see additional information below). | High | Very high | Low | Moderate |
Increase in suspended sediment [Show more]Increase in suspended sedimentBenchmark. An arbitrary short-term, acute change in background suspended sediment concentration e.g., a change of 100 mg/l for one month. The resultant light attenuation effects are addressed under turbidity, and the effects of rapid settling out of suspended sediment are addressed under smothering. Further details EvidenceThe abundance of most bryozoan species declines with increasing suspended sediment loads. Bryozoans are suspension feeding organisms that may be adversely affected by increases in suspended sediment, due to clogging of their feeding apparatus. The abundance of bryozoans is positively correlated with supply of hard substrata and hence with current strength as strong currents decrease the potential for siltation (Eggleston, 1972b; Ryland, 1976). However, Conopeum reticulum has been recorded on stones and boulders, around which fine sediments tend to collect. In addition, Conopeum reticulum occurs in estuarine waters, such as the higher reaches of the River Tamar, Plymouth which, while below the turbidity maxima, are probably of higher turbidity and suspended sediment loads than coastal waters. However, in areas of siltation it may be restricted to vertical or steep surfaces. Therefore, Conopeum reticulum may be more tolerant of siltation and suspended sediment than most encrusting bryozoans, and an intolerance of low has been suggested at the benchmark level. | Low | Immediate | Not sensitive | Low |
Decrease in suspended sediment [Show more]Decrease in suspended sedimentBenchmark. An arbitrary short-term, acute change in background suspended sediment concentration e.g., a change of 100 mg/l for one month. The resultant light attenuation effects are addressed under turbidity, and the effects of rapid settling out of suspended sediment are addressed under smothering. Further details EvidenceA decrease in suspended sediment may reduce the availability of organic particulates. However, a decrease in particulates is likely to encourage the settlement and growth of bryozoans including Conopeum reticulum. Therefore, tolerant* has been recorded. A decrease in sediment load is also likely to allow competitors such as other bryozoans and ascidians to colonize the habitat. | Tolerant* | Not relevant | Not sensitive* | Low |
Desiccation [Show more]Desiccation
EvidenceLower shore populations of Conopeum reticulum may be adversely affected by desiccation. Conopeum reticulum is restricted to damp habitats or rockpools on the shore. Therefore, an increase in desiccation at the benchmark level, or resulting from overturning of boulders or stones to which colonies are attached, would probably kill the affected colonies. Therefore, an intolerance of intermediate has been recorded to represent loss of the populations extent in the intertidal. The subtidal is unlikely to be exposed to desiccation. Recoverability is likely to be very high (see additional information below). | Intermediate | Very high | Low | Low |
Increase in emergence regime [Show more]Increase in emergence regimeBenchmark. A one hour change in the time covered or not covered by the sea for a period of one year. Further details EvidenceAn increase in emergence will result in a larger proportion of the population being exposed to intertidal conditions, increased extremes of temperature, reduced ability to feed and an increased risk of desiccation (see above). Therefore, an intolerance of intermediate has been recorded. Recoverability is likely to be very high (see additional information below). | Intermediate | Very high | Low | Low |
Decrease in emergence regime [Show more]Decrease in emergence regimeBenchmark. A one hour change in the time covered or not covered by the sea for a period of one year. Further details EvidenceA decrease in emergence, at the benchmark level, is likely to provide additional habitat for colonization by Conopeum reticulum as well as other bryozoans and epifauna, and reduce the risk of desiccation to existing colonies. Therefore, tolerant* has been recorded. | Tolerant* | Not relevant | Not sensitive* | Low |
Increase in water flow rate [Show more]Increase in water flow rateA change of two categories in water flow rate (view glossary) for 1 year, for example, from moderately strong (1-3 knots) to very weak (negligible). Further details EvidenceConopeum reticulum has been reported in strong to weak tidal streams (JNCC, 1999). Therefore, it is probably tolerant of a wide range of water flow. However, an increase in water flow from e.g. moderately strong to very strong may interfere with larval settlement and remove shells and small stones, to which colonies were attached. Therefore, a proportion of the population may be lost and an intolerance of intermediate has been recorded. Recoverability is likely to be very high (see additional information below). | Intermediate | Very high | Low | Low |
Decrease in water flow rate [Show more]Decrease in water flow rateA change of two categories in water flow rate (view glossary) for 1 year, for example, from moderately strong (1-3 knots) to very weak (negligible). Further details EvidenceConopeum reticulum has been recorded from sites subject to strong to weak tidal streams (JNCC, 1999). The abundance of bryozoans is positively correlated with supply of hard substrata and hence with current strength as strong currents (Eggleston, 1972b; Ryland, 1976). Bryozoans are active suspension feeders, however, their feeding currents are probably fairly localized and they are dependent on water flow to bring adequate food supplies within reach (McKinney, 1986). A decrease in water flow to very weak, in the absence of compensatory wave action, is likely to reduce food availability and increase the risk of siltation. Therefore, a proportion of the population may be lost and an intolerance of intermediate has been recorded. Recoverability is likely to be very high (see additional information below). | Intermediate | Very high | Low | Low |
Increase in temperature [Show more]Increase in temperature
For intertidal species or communities, the range of temperatures includes the air temperature regime for that species or community. Further details EvidenceMenon (1972, 1974) reported Conopeum reticulum acclimated to temperatures between 6 to 22 °C and that increasing temperature increased growth and feeding rate but reduced zooid size. Acclimation to increased temperatures increased the median lethal temperature. Menon (1972) reported an upper lethal temperature of 30 °C in colonies acclimated to 22 °C and 32 °C in colonies grown at 22 °C. Therefore, it is likely that colonies will tolerate higher temperatures in the summer months than in the winter months. However, Menon (1972) noted that colonies kept at 6 or 22 °C in culture did not reach sexual maturity. Overall, Conopeum reticulum exhibited the highest temperature tolerance of the bryozoans studied by Menon (1972). Conopeum reticulum is probably widely distributed around the British Isles and into the Mediterranean, and occurs in the Cochin backwaters of the southwest coast of India (Menon, 1973). Therefore, it is probably tolerant of long term change in temperatures in British waters. It would probably also tolerate acute temperature change at the benchmark level. Therefore, an intolerance of low has been recorded, to represent the effects of temperature on growth and reproduction. | Low | Immediate | Not sensitive | Moderate |
Decrease in temperature [Show more]Decrease in temperature
For intertidal species or communities, the range of temperatures includes the air temperature regime for that species or community. Further details EvidenceMenon (1972) reported that colonies acclimated to 6 and 12 °C survived below zero, polypides only dying at ca -1.8 °C when ice crystals appear in seawater. Polypides of colonies acclimated to higher temperatures died above zero, e.g. at 2.5 °C in colonies acclimated to 22 °C. Therefore, colonies are probably more tolerant of low temperatures in winter than summer. However, Menon (1972) noted that colonies kept at 6 or 22 °C in culture did not reach sexual maturity. Eggleston (1972a) noted that the unusually cold winter of 1962/63 delayed the onset of reproduction in some species of bryozoan by up to 2 months. However, Ryland (1970) suggested that temperature was but one factor controlling summer growth and reproduction in temperate bryozoans. | Low | Immediate | Not sensitive | Moderate |
Increase in turbidity [Show more]Increase in turbidity
EvidenceAn increase in turbidity will decrease light penetration, and hence primary productivity, potentially decreasing the food available to Conopeum reticulum. However, Conopeum reticulum would probably be able to feed on organic particulates. It may be more intolerant in the summer months, when lack of food would reduce growth and reproduction. In addition, larval growth may be delayed, and hence larval mortality increased. Therefore, an intolerance of low has been recorded. | Low | Immediate | Not sensitive | Low |
Decrease in turbidity [Show more]Decrease in turbidity
EvidenceA decrease in turbidity is likely to result in an increase in primary productivity and phytoplankton availability. Therefore, tolerant has been recorded. | Tolerant | Not relevant | Not sensitive | Not relevant |
Increase in wave exposure [Show more]Increase in wave exposureA change of two ranks on the wave exposure scale (view glossary) e.g., from Exposed to Extremely exposed for a period of one year. Further details EvidenceConopeum reticulum has been recorded from wave exposed to extremely wave sheltered habitats (JNCC, 1999). Its encrusting habit may allow this species to survive in areas of greater wave exposure. However, its intolerance will probably depend on its substratum. Colonies on stable hard substrata such as bedrock will probably survive, whereas colonies of boulders, rocks or cobbles are likely to be destroyed by rolling or overturning. In addition, colonies on stones on sediment may be destroyed by additional sediment abrasion. Therefore, an increase in wave exposure from sheltered to exposed will probably not adversely affect the population, whereas an increase from moderately exposed to very exposed may result in loss of a proportion of the population, depending on substratum, and an intolerance of intermediate has been recorded. Recoverability is likely to be very high (see additional information below). | Intermediate | Very high | Low | Low |
Decrease in wave exposure [Show more]Decrease in wave exposureA change of two ranks on the wave exposure scale (view glossary) e.g., from Exposed to Extremely exposed for a period of one year. Further details EvidenceConopeum reticulum is recorded from wave exposed to extremely wave sheltered habitats. A further decrease in wave exposure may increase the risk of deoxygenation (stagnant conditions) or siltation (see above) unless the tidal streams were sufficient to ensure adequate water exchange. Suspension feeding organisms are reduced in abundance or absent from areas with little water movement (either due to currents or wave action or both). Therefore, in areas of sufficient water flow due to currents a further reduction in wave exposure may have negligible effects. Therefore, an intolerance of low has been recorded. Populations in areas subject to only weak or negligible current are likely to be more intolerant. | Low | Immediate | Not sensitive | Low |
Noise [Show more]Noise
EvidenceThe species is unlikely to be sensitive to changes in noise vibrations. | Tolerant | Not relevant | Not sensitive | High |
Visual presence [Show more]Visual presenceBenchmark. The continuous presence for one month of moving objects not naturally found in the marine environment (e.g., boats, machinery, and humans) within the visual envelope of the species or community under consideration. Further details EvidenceThe species is unlikely to be sensitive to changes in visual perception. | Tolerant | Not relevant | Not sensitive | High |
Abrasion & physical disturbance [Show more]Abrasion & physical disturbanceBenchmark. Force equivalent to a standard scallop dredge landing on or being dragged across the organism. A single event is assumed for assessment. This factor includes mechanical interference, crushing, physical blows against, or rubbing and erosion of the organism or habitat of interest. Where trampling is relevant, the evidence and trampling intensity will be reported in the rationale. Further details. EvidenceConopeum reticulum has been recorded on boulders, rocks and stones on sediment (JNCC, 1999). Therefore, it is probably tolerant of some sediment scour. However, it is likely that Conopeum reticulum colonies on stones and rocks, and to some extent boulders, are probably ephemeral, being removed by scour by increased wave action due to winter storms. Abrasion by a passing anchor is likely to roll or overturn stones, to which colonies are attached, and damage but not remove colonies on bedrock. A passing scallop dredge is likely to damage but not remove colonies, unless they are removed with rocks to which they are attached (see substratum loss). Overall, an intolerance of intermediate has been recorded, although recoverability is probably very high (see additional information below). | Intermediate | Very high | Low | Very low |
Displacement [Show more]DisplacementBenchmark. Removal of the organism from the substratum and displacement from its original position onto a suitable substratum. A single event is assumed for assessment. Further details EvidenceConopeum reticulum may be displaced together with the rocks or cobbles to which they are attached. If they are displaced to suitable habitats they will probably survive as long as they were not crushed in the process. However, removal of a colony from its substratum would probably be fatal, and encrusting bryozoa are not known to be able to reattach. Therefore, an intolerance of high has been recorded. Recoverability is likely to be very high (see additional information below). | High | Very high | Low | High |
Chemical pressures
Use [show more] / [show less] to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Synthetic compound contamination [Show more]Synthetic compound contaminationSensitivity is assessed against the available evidence for the effects of contaminants on the species (or closely related species at low confidence) or community of interest. For example:
The evidence used is stated in the rationale. Where the assessment can be based on a known activity then this is stated. The tolerance to contaminants of species of interest will be included in the rationale when available; together with relevant supporting material. Further details. EvidenceBryozoans are common members of the fouling community, and amongst those organisms most resistant to antifouling measures, such as copper-containing anti-fouling paints (Soule & Soule, 1977; Holt et al., 1995). Bryan & Gibbs (1991) reported that there was little evidence regarding TBT toxicity in bryozoa with the exception of the encrusting Schizoporella errata, which suffered 50% mortality when exposed for 63 days to 100ng/l TBT. Rees et al. (2001) reported that the abundance of epifauna (including bryozoans) had increased in the Crouch estuary in the five years since TBT was banned from use on small vessels. This last report suggests that bryozoans may be at least inhibited by the presence of TBT. Hoare & Hiscock (1974) suggested that Polyzoa (Bryozoa) were amongst the most sensitive species to acidified halogenated effluents in Amlwch Bay, Anglesey but did not record Conopeum spp. in their survey. Overall, an intolerance of intermediate has been recorded to represent the likely intolerance of bryozoans to synthetic contaminants. Recoverability is likely to be very high (see additional information below). | Intermediate | Very high | Low | Very low |
Heavy metal contamination [Show more]Heavy metal contaminationEvidenceBryozoans are common members of the fouling community, and amongst those organisms most resistant to antifouling measures, such as copper-containing anti-fouling paints (Soule & Soule, 1977; Holt et al., 1995). Bryozoans were shown to bioaccumulate heavy metals to a certain extent (Holt et al., 1995). For example Bowerbankia gracilis and Nolella pusilla accumulated Cd, exhibiting sublethal effects (reduced sexual reproduction and inhibited resting spore formation) between 10-100 µg Cd /l and fatality above 500 µg Cd/l (Kayser, 1990). However, given the tolerance of bryozoans to copper based anti-fouling treatments, and assuming similar physiology between species, an intolerance of low has been recorded albeit with very low confidence. | Low | Immediate | Not sensitive | Very low |
Hydrocarbon contamination [Show more]Hydrocarbon contaminationEvidenceLittle information on the effects of hydrocarbons on bryozoans was found. Houghton et al. (1996) reported a reduction in the abundance of intertidal encrusting Bryozoa (no species given) at oiled sites after the Exxon Valdez oil spill. Soule & Soule (1979) reported that the encrusting bryozoan Membranipora villosa was not found in the impacted area for 7 months after the December 1976 Bunker C oil spill in Los Angeles Harbour. Of the eight species of bryozoan recorded on the nearby breakwater two weeks after the incident, only three were present in April and by June all had been replaced by dense growths of the erect bryozoan Scrupocellaria diegensis. Mohammad (1974) reported that Bugula spp. and Membranipora spp. were excluded from settlement panels near a Kuwait Oil terminal subject to minor but frequent oil spills. Encrusting bryozoans are also probably intolerant of the smothering effects of oil pollution, resulting in suffocation of colonies. Therefore, given the above evidence of intolerance in other Membraniporidae, a intolerance of high has been recorded, albeit at low confidence. Recoverability is probably very high (see additional information below). | High | Very high | Low | Low |
Radionuclide contamination [Show more]Radionuclide contaminationEvidenceInsufficient | No information | Not relevant | No information | Not relevant |
Changes in nutrient levels [Show more]Changes in nutrient levelsEvidenceA moderate increase in nutrient levels may increase the food available to Conopeum reticulum, either in the form of phytoplankton or detritus. Jakola & Gulliksen (1987) reported that encrusting bryozoans were excluded from the vicinity of a sewage outfall from Tromsö, Norway. However, they suggested that the effect was primarily due to sedimentation. Little other information on the effects of nutrients enrichment on bryozoans were found. | No information | Not relevant | No information | Not relevant |
Increase in salinity [Show more]Increase in salinity
EvidenceConopeum reticulum may be found intertidally in damp locations, under boulders and in rockpools. It may, therefore be exposed to increased salinity due to evaporation and is probably more tolerant than subtidal bryozoans. However, no information sufficient to make an assessment was found. | No information | Not relevant | No information | Not relevant |
Decrease in salinity [Show more]Decrease in salinity
EvidenceConopeum reticulum is found in marine and estuarine waters and is considered to be euryhaline, although Ryland (1970) noted that its estuarine distribution may be inaccurate due to common confusion with Conopeum seurati and Electra crustulenta. However, Conopeum reticulum was abundant in the higher reaches of the Crouch and Tamar estuaries at salinities of 21- 32psu (Cook, 1964; Hayward & Ryland, 1998). Menon (1973) reported that Conopeum reticulum occurred in the outer reaches of the Cochin backwaters in southern India, which were affected by significant freshwater runoff during the monsoon season. In the Cochin backwaters Conopeum reticulum was more abundant in the shallow subtidal (1-2m) and absent from the intertidal. | Low | Immediate | Not sensitive | Low |
Changes in oxygenation [Show more]Changes in oxygenationBenchmark. Exposure to a dissolved oxygen concentration of 2 mg/l for one week. Further details. EvidenceLittle information concerning the effects of hypoxia on bryozoans was found. Sagasti et al. (2000) reported that epifauna communities, including dominant species such as Conopeum tenuissimum and Membranipora tenuis, were unaffected by periods of moderate hypoxia (ca 0.35 -1.4 ml/l) and short periods of hypoxia (<0.35 ml/l) in the York River, Chesapeake Bay. Therefore, assuming similar physiology between species of Conopeum, an intolerance of low has been recorded. | Low | Immediate | Not sensitive | Moderate |
Biological pressures
Use [show more] / [show less] to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Introduction of microbial pathogens/parasites [Show more]Introduction of microbial pathogens/parasitesBenchmark. Sensitivity can only be assessed relative to a known, named disease, likely to cause partial loss of a species population or community. Further details. EvidenceNo information found | No information | Not relevant | No information | Not relevant |
Introduction of non-native species [Show more]Introduction of non-native speciesSensitivity assessed against the likely effect of the introduction of alien or non-native species in Britain or Ireland. Further details. EvidenceNo information found | No information | Not relevant | No information | Not relevant |
Extraction of this species [Show more]Extraction of this speciesBenchmark. Extraction removes 50% of the species or community from the area under consideration. Sensitivity will be assessed as 'intermediate'. The habitat remains intact or recovers rapidly. Any effects of the extraction process on the habitat itself are addressed under other factors, e.g. displacement, abrasion and physical disturbance, and substratum loss. Further details. EvidenceConopeum reticulum is unlikely to be subject to specific extraction. | Not relevant | Not relevant | Not relevant | Not relevant |
Extraction of other species [Show more]Extraction of other speciesBenchmark. A species that is a required host or prey for the species under consideration (and assuming that no alternative host exists) or a keystone species in a biotope is removed. Any effects of the extraction process on the habitat itself are addressed under other factors, e.g. displacement, abrasion and physical disturbance, and substratum loss. Further details. EvidenceConopeum reticulum is not known to be associated with species or habitats subject to extraction. | Not relevant | Not relevant | Not relevant | Not relevant |
Additional information
Recoverability. Conopeum reticulum has a planktonic larva with a protracted life in the plankton and potentially extended dispersal. The larvae are present in estuarine and coastal waters between July and September. Conopeum reticulum can also colonize a wide variety of substrata and is a member of fouling communities. For example, Hatcher (1998) reported that Conopeum reticulum colonized artificial reef surfaces within 6 months from May to October 1991 (Hatcher, 1998). Therefore, it is likely to be able to colonize new habitats or free space rapidly, probably in 6 months or less. It grows and probably matures quickly, within a year or less, and subsequent expansion of the population and recovery of abundance, aided by the proximity of breeding colonies is also likely to occur rapidly, possibly within a few years at most.
Importance review
Policy/legislation
- no data -
Status
| National (GB) importance | - | Global red list (IUCN) category | - |
Non-native
| Parameter | Data |
|---|---|
| Native | - |
| Origin | - |
| Date Arrived | - |
Importance information
Bryozoans are grazed by sea urchins such as Echinus esculentus and Psammechinus miliaris in the subtidal. Bryozoans are also preyed on by pycnogonids (sea spiders) and nudibranchs (sea slugs) (Ryland, 1976).Bibliography
Bayer, M.M., Cormack, R.M. & Todd, C.D., 1994. Influence of food concentration on polypide regression in the marine bryozoan Electra pilosa (L.) (Bryozoa: Cheilostomata). Journal of Experimental Marine Biology and Ecology, 178, 35-50.
Best, M.A. & Thorpe, J.P., 1994. An analysis of potential food sources available to intertidal bryozoans in Britain. In Proceedings of the 9th International Bryozoology conference, Swansea, 1992. Biology and Palaeobiology of Bryozoans (ed. P.J. Hayward, J.S. Ryland & P.D. Taylor), pp. 1-7. Fredensborg: Olsen & Olsen.
Bryan, G.W. & Gibbs, P.E., 1991. Impact of low concentrations of tributyltin (TBT) on marine organisms: a review. In: Metal ecotoxicology: concepts and applications (ed. M.C. Newman & A.W. McIntosh), pp. 323-361. Boston: Lewis Publishers Inc.
Cook, P.L., 1964. The development of Electra monostachys (Busk) and Conopeum reticulum (Linnaeus), Polyzoa, Anasca. Cahiers de Biologie Marine, 5, 391-397.
Eggleston, D., 1972a. Patterns of reproduction in marine Ectoprocta off the Isle of Man. Journal of Natural History, 6, 31-38.
Eggleston, D., 1972b. Factors influencing the distribution of sub-littoral ectoprocts off the south of the Isle of Man (Irish Sea). Journal of Natural History, 6, 247-260.
Grant, A. & Hayward, P.J. 1985. Bryozoan benthic assemblages in the English Channel. In Bryozoa: Ordovician to Recent (ed. C. Nielsen & G.P. Larwood), pp. 115-124. Fredensborg: Olsen & Olsen.
Hatcher, A.M., 1998. Epibenthic colonization patterns on slabs of stabilised coal-waste in Poole Bay, UK. Hydrobiologia, 367, 153-162.
Hayward, P., Nelson-Smith, T. & Shields, C. 1996. Collins pocket guide. Sea shore of Britain and northern Europe. London: HarperCollins.
Hayward, P.J. & Ryland, J.S. (ed.) 1995b. Handbook of the marine fauna of North-West Europe. Oxford: Oxford University Press.
Hayward, P.J. & Ryland, J.S. 1998. Cheilostomatous Bryozoa. Part 1. Aeteoidea - Cribrilinoidea. Shrewsbury: Field Studies Council. [Synopses of the British Fauna, no. 10. (2nd edition)]
Hincks, T., 1880. A history of British marine Polyzoa, vol. I & II. London: John van Voorst.
Hoare, R. & Hiscock, K., 1974. An ecological survey of the rocky coast adjacent to the effluent of a bromine extraction plant. Estuarine and Coastal Marine Science, 2 (4), 329-348.
Holt, T.J., Jones, D.R., Hawkins, S.J. & Hartnoll, R.G., 1995. The sensitivity of marine communities to man induced change - a scoping report. Countryside Council for Wales, Bangor, Contract Science Report, no. 65.
Houghton, J.P., Lees, D.C., Driskell, W.B., Lindstrom & Mearns, A.J., 1996. Recovery of Prince William Sound intertidal epibiota from Exxon Valdez oiling and shoreline treatments, 1989 through 1992. In Proceedings of the Exxon Valdez Oil Spill Symposium. American Fisheries Society Symposium, no. 18, Anchorage, Alaska, USA, 2-5 February 1993, (ed. S.D. Rice, R.B. Spies, D.A., Wolfe & B.A. Wright), pp.379-411.
Howson, C.M. & Picton, B.E., 1997. The species directory of the marine fauna and flora of the British Isles and surrounding seas. Belfast: Ulster Museum. [Ulster Museum publication, no. 276.]
Hunter, E. & Hughes, R.N., 1994. Influence of temperature, food ration and genotype on zooid size in Celleporella hyalina (L.). In Proceedings of the 9th International Bryozoology Conference, Swansea, 1992. Biology and Palaeobiology of Bryozoans (ed. P.J. Hayward, J.S. Ryland & P.D. Taylor), pp. 83-86. Fredensborg: Olsen & Olsen.
Hyman, L.V., 1959. The Invertebrates, vol. V. Smaller coelomate groups. New York: McGraw-Hill.
Jakola, K.J. & Gulliksen, B., 1987. Benthic communities and their physical environment to urban pollution from the city of Tromso, Norway. Sarsia, 72, 173-182.
Jebram, D., 1970. Preliminary experiments with Bryozoa in a simple apparatus for producing continuous water currents. Helgolander Wissenschaftliche Meeresuntersuchungen, 20, 278-292.
JNCC (Joint Nature Conservation Committee), 1999. Marine Environment Resource Mapping And Information Database (MERMAID): Marine Nature Conservation Review Survey Database. [on-line] http://www.jncc.gov.uk/mermaid
McKinney, F.K., 1986. Evolution of erect marine bryozoan faunas: repeated success of unilaminate species The American Naturalist, 128, 795-809.
Menon, N.R., 1972. Heat tolerance, growth and regeneration in three North Sea bryozoans exposed to different constant temperatures. Marine Biology, 15, 1-11.
Menon, N.R., 1973. Vertical and horizontal distribution of fouling Bryozoans in Cochin backwaters, southwest coast of India. In Living and fossil Bryozoa (ed. G.P. Larwood), pp. 153-164. New York: Academic Press.
Menon, N.R., 1974. Clearance rates of food suspension and food passage rates as a function of temperature in two North Sea bryozoans. Marine Biology, 24, 65-67.
Mohammad, M-B.M., 1974. Effect of chronic oil pollution on a polychaete. Marine Pollution Bulletin, 5, 21-24.
Rees, H.L., Waldock, R., Matthiessen, P. & Pendle, M.A., 2001. Improvements in the epifauna of the Crouch estuary (United Kingdom) following a decline in TBT concentrations. Marine Pollution Bulletin, 42, 137-144. DOI https://doi.org/10.1016/S0025-326X(00)00119-3
Ryland, J.S., 1967. Polyzoa. Oceanography and Marine Biology: an Annual Review, 5, 343-369.
Ryland, J.S., 1970. Bryozoans. London: Hutchinson University Library.
Ryland, J.S., 1976. Physiology and ecology of marine bryozoans. Advances in Marine Biology, 14, 285-443.
Sagasti, A., Schaffner, L.C. & Duffy, J.E., 2000. Epifaunal communities thrive in an estuary with hypoxic episodes. Estuaries, 23 (4), 474-487.
Soule, D.F. & Soule, J.D., 1979. Bryozoa (Ectoprocta). In Hart, C.W. & Fuller, S.L.H. (eds), Pollution ecology of estuarine invertebrates. New York: Academic Press, pp. 35-76.
Winston, J.E., 1977. Feeding in marine bryozoans. In Biology of Bryozoans (ed. R.M. Woollacott & R.L. Zimmer), pp. 233-271.
Datasets
Bristol Regional Environmental Records Centre, 2017. BRERC species records recorded over 15 years ago. Occurrence dataset: https://doi.org/10.15468/h1ln5p accessed via GBIF.org on 2018-09-25.
Centre for Environmental Data and Recording, 2018. Ulster Museum Marine Surveys of Northern Ireland Coastal Waters. Occurrence dataset https://www.nmni.com/CEDaR/CEDaR-Centre-for-Environmental-Data-and-Recording.aspx accessed via NBNAtlas.org on 2018-09-25.
Fenwick, 2018. Aphotomarine. Occurrence dataset http://www.aphotomarine.com/index.html Accessed via NBNAtlas.org on 2018-10-01
Kent Wildlife Trust, 2018. Biological survey of the intertidal chalk reefs between Folkestone Warren and Kingsdown, Kent 2009-2011. Occurrence dataset: https://www.kentwildlifetrust.org.uk/ accessed via NBNAtlas.org on 2018-10-01.
Kent Wildlife Trust, 2018. Kent Wildlife Trust Shoresearch Intertidal Survey 2004 onwards. Occurrence dataset: https://www.kentwildlifetrust.org.uk/ accessed via NBNAtlas.org on 2018-10-01.
Merseyside BioBank., 2018. Merseyside BioBank (unverified). Occurrence dataset: https://doi.org/10.15468/iou2ld accessed via GBIF.org on 2018-10-01.
National Trust, 2017. National Trust Species Records. Occurrence dataset: https://doi.org/10.15468/opc6g1 accessed via GBIF.org on 2018-10-01.
NBN (National Biodiversity Network) Atlas. Available from: https://www.nbnatlas.org.
Norfolk Biodiversity Information Service, 2017. NBIS Records to December 2016. Occurrence dataset: https://doi.org/10.15468/jca5lo accessed via GBIF.org on 2018-10-01.
OBIS (Ocean Biodiversity Information System), 2024. Global map of species distribution using gridded data. Available from: Ocean Biogeographic Information System. www.iobis.org. Accessed: 2024-05-19
South East Wales Biodiversity Records Centre, 2018. SEWBReC Marine and other Aquatic Invertebrates (South East Wales). Occurrence dataset:https://doi.org/10.15468/zxy1n6 accessed via GBIF.org on 2018-10-02.
Citation
This review can be cited as:
Last Updated: 13/08/2005