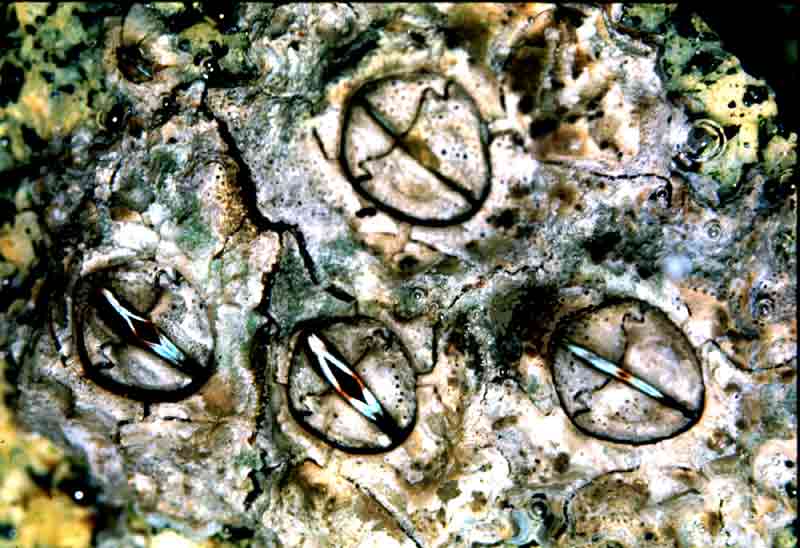
Poli's stellate barnacle (Chthamalus stellatus)
Distribution data supplied by the Ocean Biodiversity Information System (OBIS). To interrogate UK data visit the NBN Atlas.Map Help
| Researched by | Karen Riley | Refereed by | Prof. Alan J. Southward |
| Authority | (Poli, 1791) | ||
| Other common names | - | Synonyms | - |
Summary
Description
An intertidal barnacle with six solid wall plates, an oval-shaped operculum opening, and a membranous base. Juveniles have a kite-shaped opercular opening. The rostral plate is relatively narrow, plates are of roughly equal size, and its rostral plate is not fused with the rostrolateral plates. Usually conical in shape, however when crowded may become tubular. It may reach a diameter of approximately 14 mm, depending on habitat, food availability and level on the shore. The tissue inside the opercular aperture is bright blue with black and orange markings.
Recorded distribution in Britain and Ireland
A southern, warm-water species recorded on the south and west coasts of the British Isles as far north as the Shetland Isles. The species is less abundant along the channel towards The Isle of Wight, its eastern limit.
Global distribution
Occurs in The Black Sea, along most of the Mediterranean coast, and further south in Tunisia, Madeira, the Azores, and Cape Verde Islands. No recorded exist further south of this point.
Habitat
Recorded in the mid to low eulittoral zone on exposed rocky shores. Its vertical distribution overlaps with Chthamalus montagui and Semibalanus balanoides.
Depth range
Not relevantIdentifying features
- Shell wall of six solid plates.
- Oval-shaped operculum opening.
- Joint between the terga and scuta crosses the centre line at one third or more of the distance towards the rostrum.
- Tissue inside the operculum opening is usually bright blue with brown and orange markings.
- Junction between the terga and scuta is convex towards rostral plate.
- Shell base is membranous.
Additional information
Before 1976 Chthamalus montagui was considered a variety of Chthamalus stellatus, but in 1976 was identified as a distinct species, due to differences in its vertical zonation on the shore and morphology, particularly in the shape of the opercular plates, setation of the smaller cirri. (Southward, 1976).
Listed by
- none -
Biology review
Taxonomy
| Level | Scientific name | Common name |
|---|---|---|
| Phylum | Arthropoda | Arthropods, joint-legged animals, e.g. insects, crustaceans & spiders |
| Family | Chthamalidae | |
| Genus | Chthamalus | |
| Authority | (Poli, 1791) | |
| Recent Synonyms | ||
Biology
| Parameter | Data | ||
|---|---|---|---|
| Typical abundance | High density | ||
| Male size range | Up to 1.4 cm | ||
| Male size at maturity | |||
| Female size range | Small(1-2cm) | ||
| Female size at maturity | |||
| Growth form | |||
| Growth rate | ca. 10-55 | ||
| Body flexibility | None (less than 10 degrees) | ||
| Mobility | Sessile, permanent attachment | ||
| Characteristic feeding method | Active suspension feeder, See additional information | ||
| Diet/food source | Planktotroph | ||
| Typically feeds on | Generally feeds on plankton. | ||
| Sociability | Gregarious | ||
| Environmental position | Epifaunal | ||
| Dependency | Independent. | ||
| Supports | None | ||
| Is the species harmful? | No | ||
Biology information
Feeding. Chthamalus stellatus / Chthamalus montagui generally feed on small plankton. They can consume diatoms but were found not to grow under a regime dominated by diatoms (Barnes & Barnes, 1965). Normal feeding of chthamalids involves a cirral beat. This cirral beat is also noted to be a respiratory mechanism (Anderson & Southward, 1987). However, in high wave exposure they tend to hold their cirri out stiffly against the water current for a long period of time, retracting when food is captured (Crisp, 1950). Barnacles living in wave exposed conditions may benefit from this passive suspension feeding habit where cirral beating and consequent energy expenditure are minimised (Crisp, 1950). Rates of cirral beat decrease with age and size, but increase with temperature (Anderson & Southward, 1987). Green (1961) reported that barnacles higher up on shore had a higher cirral beat frequency than those at lower levels. However, Southward (1955; 1964(b)) found no similar trends. Southward (1955) found that there was no response of Chthamalus stellatus or Chthamalus montagui in still water and that cirral beating was only induced at a current of approximately 10 cm/sec. The extension response was also sometimes shown. The cirral beating frequency is also related to temperature, shown by experiments by Southward (1955). Chthamalus stellatus / Chthamalus montagui barnacles kept at a temperature of 0 °C did not react to touch after an hour. He also found that they remained inactive at a temperature up to 5 °C. Between 5 and 30 °C there was a linear increase to 10 beats every 10 seconds. This slowly declined above 33 °C and dropped rapidly at 36 °C. Although the species resisted coma above a temperature of 40 °C, all cirral beating ceased at 37.5 °C.
Respiration. Sessile barnacles have a pair of gills: pleats of the mantle wall, attached to the mantle cavity (Stubbings, 1975). Rainbow (1984) also stated that the cirri might also play an important role in respiration. There is usually a slow respiratory pumping beat, with varied emergence of the cirri.
Moulting. Barnacles need to moult in order to grow. Feeding rate and temperature determine the frequency of moulting. Moulting does not take place during winter when phytoplankton levels and temperatures are low (Crisp & Patel, 1960).
Growth. Once the barnacle is fixed in place it is unable to detach again (Crisp, 1955). All species grow faster in early life and slower in later life, and chthamalids tend to become tubular when crowded (Southward & Crisp, 1965). The growth rate varies with a variety of biological and environmental factors, including current flow, orientation with respect to current, food supply, wave exposure, shore height, surface contour, and intra- or inter-specific competition. Growth in Chthamalus spp. takes place along the whole internal surface of the one-layered plates (Bourget, 1977). The growth rate for Chthamalus stellatus / Chthamalus montagui has been reported by Barnes (1956; Crisp & Bourget (1985) as between 10 - 55 µm per day (relatively slow) in the linear phase. Crisp (1950) noticed that Chthamalus stellatus / Chthamalus montagui reached a maximum size of 0.2 to 1.4 cm. Chthamalus stellatus / Chthamalus montagui was found to have a lower growth rate than many other species of barnacles (Relini, 1983). The species reached a basal diameter of 2-2.5 mm in 3 months, 3.5-4 one year later, up to 8 mm in the 2nd year of growth, but generally no more than about 5-6 mm (Relini, 1983). Sometimes a decrease in size was noticeable, due to abrasion. This low growth rate was found to be associated with a low metabolic rate, or low oxygen consumption, by Barnes & Barnes (1965). Benedetti-Cecchi et al. (2000) observed that Chthamalus stellatus barnacles in the northwest Mediterranean were significantly larger the higher up shore that they were found. However, no significant difference in growth rate was noted by Benedetti-Cecchi et al. (2000), with the growth rate of juveniles being between 0.4 and 0.8 mm per year and greater mortality of young and adults in a low shore environment.
Parasites and epizoites. Healy (1986, in O'Riordan et al., 1992) observed the parasitic isopod, Hemioniscus balani in Chthamalus stellatus and Chthamalus montagui in Ireland, although it was never present in Lough Hyne populations. However, Southward & Crisp (1954) found that although it attacks and sterilises Semibalanus balanoides individuals, it does not normally attack chthamalids on British shores.
Further Information. The dog whelk, Nucella lapillus, feeds on barnacles. The species of Chthamalus spp. are less at risk of this due to their smaller size in comparison with Semibalanus balanoides, but nevertheless, it can still have a negative impact on their abundance. Other predators which pull shells or cirri of barnacles off the rock include crabs, amphipods, shore fish such as wrasse and sometimes herring gulls (Moore & Kitching, 1939), in particular, the shanny Blennius pholis (Kendall & Bedford, 1987). Another possible predator is the polychaete, Eulalia viridis (Moore & Kitching, 1939). Chthamalus spp. is also known to be displaced by Patella spp. and smothered by Mytilus spp. and algae at lower shore levels (Moore & Kitching, 1939). Empty barnacle cases provide homes for small periwinkles, , small bivalves and the isopod, Campecopea hirsuta (Fish & Fish, 1996). In order to protect themselves from changes in temperature, desiccation and a lowering of salinity, intertidal barnacles are usually able to close their aperture tightly (Moore & Kitching, 1939).
Habitat preferences
| Parameter | Data |
|---|---|
| Physiographic preferences | Open coast |
| Biological zone preferences | Lower eulittoral, Mid eulittoral |
| Substratum / habitat preferences | Artificial (man-made), Bedrock, Large to very large boulders |
| Tidal strength preferences | Moderately strong 1 to 3 knots (0.5-1.5 m/sec.), Strong 3 to 6 knots (1.5-3 m/sec.), Very strong > 6 knots (>3 m/sec.) |
| Wave exposure preferences | Exposed |
| Salinity preferences | Full (30-40 psu) |
| Depth range | Not relevant |
| Other preferences | No text entered |
| Migration Pattern | Non-migratory or resident |
Habitat Information
Geographical distribution. Crisp et al. (1981) described the distribution of Chthamalus stellatus and Chthamalus montagui. Chthamalus stellatus is abundant along the western coasts of Britain and Ireland. It does not occur in the central Irish Sea. It occurs in northern and north-eastern Scotland including at exposed locations in Shetland. In France, it is abundant westwards from Roscoff, is absent from the Bay of St. Malo, and is less common east of Roscoff. Chthamalus stellatus occurs along Irish coasts from Antrim around to Wexford, although it only occurs in abundance along the west coast. The species is less abundant along the channel towards the Isle of Wight, its eastern limit. Individuals recorded as Chthamalus stellatus in the Indian and Pacific Oceans are, in fact, other species of the stellatus group.
Vertical zonation. Chthamalus stellatus is dominant over Chthamalus montagui at exposed sites (Southward, 1976; Crisp et al., 1981). Where the species overlap, Chthamalus montagui has a greater vertical distribution, extending above and below that of Chthamalus stellatus (Burrows et al., 1992) and, while Chthamalus montagui is more common between MHWS & MHWN, Chthamalus stellatus is abundant lower down at MTL and below (Pannacciulli & Relini, 2000). Chthamalus stellatus inhabits the lower half of the intertidal, but in wave-exposed or wet and shady places they occur higher up on shore (Crisp et al., 1981; Pannacciulli & Relini, 2000). Physical factors such as exposure to seawater, desiccation and poor food supply limit the distribution of barnacles on the upper shore, whereas competition for space, predation and strong wave action limit the distribution at low and mid shore levels (Pannacciulli & Relini, 2000). The higher the species occurs up on the shore, the more resistant to desiccation influences they tend to be (Southward, 1955). The distribution of Chthamalus spp. is not affected by small increases in algal cover. Hawkins & Hartnoll (1982) found that the lower shore level limit was controlled by the presence of algal turf. However, rapid increases to 100 % algal cover can lead to a massive decline in barnacle populations to almost zero in a year or two (Southward, 1991).
Substratum preference. Barnacles attach themselves to hard, rough surfaces and are rarely found on chalk cliffs (Moore & Kitching, 1939). Moore & Kitching (1939) also suggested that this may be because the surface is smooth, washed away easily, or too porous (making it possible to be dried out from below).
Temperature dependence and competition. Chthamalus spp. prefer warm temperatures, whereas Semibalanus balanoides prefers low temperatures. This is reflected by the changes in their distribution with changes in climate. For example, in the severe winter of 1962-63, Chthamalus populations declined (Southward, 1967) while Semibalanus balanoides increased, and in the temperature rise of 1988-89, the trend was reversed (Southward, 1991). Long-term trends are also evident. A decline in Chthamalus populations and an increase in Semibalanus balanoides occurred between 1962 and 1980, corresponding with a decrease in sea temperatures (Southward, 1991). Since 1980 there has been a general increase in Chthamalus spp. (Southward, 1991), maybe corresponding with gradual climate warming. Southward & Crisp (1954) noted that in 1948-51, during high temperatures in the British Isles Chthamalus spp. Dominated over Semibalanus balanoides, and from 1952, during lower temperatures, there was a resurgence of Semibalanus balanoides. Southward (1991) noted a two-year phase lag between temperature trends and changes in barnacle abundance in Plymouth. Chthamalus spp. are more abundant in waters where the mean temperatures are above 10°C for several months of the year (Southward, 1955).
Life history
Adult characteristics
| Parameter | Data |
|---|---|
| Reproductive type | Gonochoristic (dioecious), Self-fertilization, Sexual |
| Reproductive frequency | Annual episodic |
| Fecundity (number of eggs) | 1,000-10,000 |
| Generation time | 1-2 years |
| Age at maturity | 9 - 10 months |
| Season | May - August |
| Life span | 2-5 years |
Larval characteristics
| Parameter | Data |
|---|---|
| Larval/propagule type | - |
| Larval/juvenile development | Planktotrophic |
| Duration of larval stage | 11-30 days |
| Larval dispersal potential | Greater than 10 km |
| Larval settlement period | Insufficient information |
Life history information
Before 1976 there was no distinction between Chthamalus stellatus and Chthamalus montagui. Since 1976 the existence of two separate species was recognised (Southward, 1976). Therefore, papers pre-1976 on Chthamalus stellatus have been recorded as for both species, below.
Fertilization. Sexual maturity of Chthamalus stellatus was attained at a rostro-carinal diameter of 4.0-6.8 mm (O'Riordan et al., 1992). A pre-1976 observation by Southward & Crisp (1954) suggests that Chthamalus stellatus is able to breed in its first year after 9 to 10 months of settlement. Sperm is activated by the oviducal gland and transferred to the oviducal sac via the penis of a neighbouring barnacle (Barnes, 1989). The barnacle penis is substantially longer than the body and is capable of searching an area around the adult to find a receptive 'functional female' (Rainbow, 1984). Fertilized egg masses (egg lamellae) are brooded in the mantle cavity (O'Riordan et al., 1995), outside the body (Barnes, 1989). Barnacles generally reproduce by cross-fertilization, but Chthamalus spp. have been shown to self-fertilize when isolated (Barnes & Barnes, 1950; Barnes & Crisp, 1956; Barnes, 1989); this usually occurs high up on shore. However, it has been noted that oviposition is delayed (Barnes & Barnes, 1950; Barnes, 1989) and the resulting eggs can be slightly abnormal and are considered less viable (Barnes, 1989).
Breeding season. The onset of the breeding season in the United Kingdom was noticed by Crisp (1950) to spread up the shore level over several months. Southward (1978) suggested that Chthamalus montagui breeds one to two months later than Chthamalus stellatus. However, Crisp et al. (1981) found little difference in SW Britain, with the main breeding peak in June and August (O'Riordan et al., 1995). Throughout the breeding season most individuals produce several broods (Burrows et al., 1992; O'Riordan et al., 1992), with a small percentage of the population remaining reproductively active throughout the year (O'Riordan et al., 1995); Barnes, 1989). After the maturation of each brood ovarian and penis re-development takes place ( O'Riordan et al., 1995; Barnes & Barnes, 1965; Barnes & Barnes, 1977; Burrows, 1988; Anderson, 1994). According to Hines (1978) temperature and food availability are the main factors controlling the duration of the breeding season and the embryonic development rate. In fact, Burrows (1988, in Kendall & Bedford, 1987) found the onset of the breeding season to be correlated with a sea temperature of 10 °C or above. Breeding of Chthamalus stellatus and Chthamalus montagui usually takes place earlier in the year in continental Europe than in the British Isles (Relini & Matricardi, 1979; Relini, 1983; Miyares, 1986, all in O'Riordan et al., 1995). Crisp (1950) suggested that for Chthamalus montagui and Chthamalus stellatus in the United Kingdom, breeding commenced earlier with decreasing longitude and easterly longitude. However, in the Mediterranean, the breeding season usually occurs in July and August (Mizrahi & Achihuv, 1990, in O'Riordan et al., 1995). Breeding of Chthamalus stellatus in France occurs in April (Barnes, 1992), and correlates with mean air and sea temperatures of 11 - 12 °C, and maximum temperatures of 14 °C. Barnes (1992) found that at an upper temperature limit of 20 to 21°C in the sea and 24 to 25 °C in the air reproductive activity decreased. Southward & Crisp (1956) noted that the interval between broods in Chthamalus stellatus and Chthamalus montagui became shorter at higher temperatures. Barnes & Barnes (1965) found that in high suspended solids and low salinity, there was a decrease in the number of eggs per brood of Chthamalus stellatus in Europe. Older barnacles are able to breed at a smaller size than younger barnacles. For instance, experiments by O'Riordan et al. (1992) showed that in their first year Chthamalus stellatus and Chthamalus montagui breed once or more, and more than once thereafter. Chthamalus stellatus or Chthamalus montagui are very tolerant of high periods of emersion, yet Patel & Crisp (1960) found that when barnacles which were brooding eggs were kept out of the water, a second batch of eggs was not produced.
Embryonic development. In both Chthamalus stellatus and Chthamalus montagui it took approximately 23 days for embryos to develop completely in vivo at 15 °C (Burrows et al., 1992), whereas Burrows (1988, in Kendall & Bedford, 1987) found that at 15 °C it took 26 days, and Achituv & Barnes (1976) reported a value of 25 days, although the temperature is not known.
Fecundity. Burrows et al. (1992) found that the number of eggs per brood of Chthamalus stellatus ranged between 1,274 - 3,391 in Britain, depending on body size and weight. It was also noted by Burrows et al. (2000) that the fecundity generally increased with lower shore levels colonized, with estimations of 1-2 broods per year at high shore levels, 2 to over three at mid shore levels, and over 2 to over 4 at low shore levels. Fecundity in protected areas such as harbours is usually lower, possibly due to increased turbidity (Barnes, 1989). However, in Archachon (France) in highly turbid waters the effect was not so noticeable, probably due to higher nutrient concentrations (Barnes, 1989).
Annual recruitment and lifespan. The lifespan of Chthamalus stellatus and Chthamalus montagui is considered to be approximately two to three years (Southward & Crisp, 1950). However, growth is more rapid and the mortality rate is greater lower down on the shore (Southward & Crisp, 1950). Annual recruitment is low (Kendall & Bedford, 1987) and they have an increased longevity (Lewis, 1964) towards the northern limits of their distribution.
Sensitivity review
The MarLIN sensitivity assessment approach used below has been superseded by the MarESA (Marine Evidence-based Sensitivity Assessment) approach (see menu). The MarLIN approach was used for assessments from 1999-2010. The MarESA approach reflects the recent conservation imperatives and terminology and is used for sensitivity assessments from 2014 onwards.
Physical pressures
Use / to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Substratum loss [Show more]Substratum lossBenchmark. All of the substratum occupied by the species or biotope under consideration is removed. A single event is assumed for sensitivity assessment. Once the activity or event has stopped (or between regular events) suitable substratum remains or is deposited. Species or community recovery assumes that the substratum within the habitat preferences of the original species or community is present. Further details EvidenceBarnacles are permanently attached to hard rough surfaces. Therefore, loss of substratum due to activities such as spoil dumping or land claim will result in loss of individuals in the area. If suitable substrata remains within the area, colonization of juvenile barnacles is possible. Intolerance is assessed as high. Recoverability is likely to be moderate (see Additional Information section below). | High | Moderate | Moderate | High |
Smothering [Show more]SmotheringBenchmark. All of the population of a species or an area of a biotope is smothered by sediment to a depth of 5 cm above the substratum for one month. Impermeable materials, such as concrete, oil, or tar, are likely to have a greater effect. Further details. EvidenceChthamalus stellatus / Chthamalus montagui have been shown to be relatively unaffected by smothering by oil. Monterosso (1930) showed experimentally that the species can survive complete smothering by petroleum jelly for approximately two months, by respiring anaerobically. Complete smothering caused by the Torrey Canyon oil spill yielded similar results; A few Semibalanus balanoides died, yet Chthamalus stellatus / Chthamalus montagui seemed unaffected, while at Booby's bay more than 90 % had managed to clear an opening in the oil film (Smith, 1968). Although oil had very little effect on individuals, it is likely that smothering by sediment can clog breathing apparatus. Recruitment to the smothered area will also be reduced. Therefore intolerance is assessed as intermediate. Recoverability is likely to be high (see Additional Information section below). | Intermediate | High | Low | Moderate |
Increase in suspended sediment [Show more]Increase in suspended sedimentBenchmark. An arbitrary short-term, acute change in background suspended sediment concentration e.g., a change of 100 mg/l for one month. The resultant light attenuation effects are addressed under turbidity, and the effects of rapid settling out of suspended sediment are addressed under smothering. Further details EvidenceBarnacles are likely to be able to tolerate a slight increase in suspended sediment concentration. A large increase in siltation to 100 mg/l for one month is likely to block breathing apparatus and impose an energetic cost of cleaning the gills. Intolerance is therefore assessed as low. Recoverability is likely to be very high as feeding and respiratory structures are likely to be clear of particles within a short space of time. | Low | Very high | Very Low | Moderate |
Decrease in suspended sediment [Show more]Decrease in suspended sedimentBenchmark. An arbitrary short-term, acute change in background suspended sediment concentration e.g., a change of 100 mg/l for one month. The resultant light attenuation effects are addressed under turbidity, and the effects of rapid settling out of suspended sediment are addressed under smothering. Further details EvidenceA decrease in suspended sediment concentration is unlikely to affect Chthamalus stellatus populations. | Tolerant | Not relevant | Not sensitive | Not relevant |
Desiccation [Show more]Desiccation
EvidenceChthamalus stellatus is a warm water species, with its northern limit of distribution in Britain. It tends to be more tolerant to desiccation than Semibalanus balanoides. The higher the species occurs up on the shore, the more resistant to desiccation influences they tend to be (Southward, 1955). Cracks and crevices offer further protection from desiccation. Southward (1958) reported an internal temperature of 28.8 °C in an air temperature of 13.7 °C. Therefore, intolerance is assessed as low. Recoverability is likely to be very high (see Additional Information section below). | Low | Very high | Very Low | Moderate |
Increase in emergence regime [Show more]Increase in emergence regimeBenchmark. A one hour change in the time covered or not covered by the sea for a period of one year. Further details EvidenceAccording to Hines (1978) temperature and food availability are the main factors controlling the duration of the breeding season and the embryonic development rate. With an increase in emergence, the period of time covered by the water would decrease, and the time available for feeding and breeding would also decrease. This is likely to reduce the growth rate and reproduction. There is also likely to be a shift downwards on the shore due to competition between Semibalanus balanoides. Intolerance is assessed as intermediate. Recoverability is likely to be high (see Additional Information section below). | Intermediate | High | Low | Moderate |
Decrease in emergence regime [Show more]Decrease in emergence regimeBenchmark. A one hour change in the time covered or not covered by the sea for a period of one year. Further details EvidenceBarnacle populations are likely to be tolerant to a decrease in emergence. With a decrease in the emergence regime, the feeding time and breeding possibilities are likely to increase. Adults of Chthamalus stellatus can survive permanent submersion (Barnes, 1953). However, competition between Semibalanus balanoides is likely to play an important role in the changes in species distribution. It is likely that the distribution of Chthamalus stellatus will move further up the shore, with no noticeable difference in the range. Intolerance is assessed as low. Recoverability is likely to be very high (see Additional Information section below). Chthamalus stellatus / Chthamalus montagui are very tolerant of high periods of emersion, yet Patel & Crisp (1960) found that when barnacles which were brooding eggs were kept out of the water, a second batch of eggs was not produced. | Low | High | Low | High |
Increase in water flow rate [Show more]Increase in water flow rateA change of two categories in water flow rate (view glossary) for 1 year, for example, from moderately strong (1-3 knots) to very weak (negligible). Further details EvidenceAn increase in water flow rate is likely to lead to a higher growth rate and annual recruitment. Intolerance is assessed as low. Recoverability is likely to be very high (see Additional Information section below). | Low | Very high | Very Low | Moderate |
Decrease in water flow rate [Show more]Decrease in water flow rateA change of two categories in water flow rate (view glossary) for 1 year, for example, from moderately strong (1-3 knots) to very weak (negligible). Further details EvidenceA decrease in the water flow rate is likely to lead to a decrease in food availability and recruitment. Intolerance is assessed as low. Recoverability is likely to be very high (see Additional Information section below). | Low | Very high | Very Low | Moderate |
Increase in temperature [Show more]Increase in temperature
For intertidal species or communities, the range of temperatures includes the air temperature regime for that species or community. Further details EvidenceChthamalus stellatus would be favoured by an increase in temperature based on the following information:
| Tolerant* | Not relevant | Not sensitive* | High |
Decrease in temperature [Show more]Decrease in temperature
For intertidal species or communities, the range of temperatures includes the air temperature regime for that species or community. Further details EvidenceDuring the severe winter of 1962-63, over the majority of the species range, they were able to withstand the cold. However, greater mortalities were noted to occur a month or two after the coldest weather (Crisp, 1964). Chthamalus spp. populations declined (Southward, 1967) while Semibalanus balanoides increased, and in the temperature rise of 1988-89 the trend was reversed (Southward, 1991). A decline in Chthamalus spp. populations and an increase in Semibalanus balanoides occurred between 1951 and 1975, corresponding with a decrease in sea temperatures (Southward, 1991). Southward & Crisp (1954) noted that in 1948-51, during high temperatures in the British Isles Chthamalus spp. dominated over Semibalanus balanoides, and during 1951-52, during lower temperatures there was a resurgence of Semibalanus balanoides. Southward (1991) noted a two year phase lag between temperature trends and changes in barnacle abundance in Plymouth. | High | Low | High | High |
Increase in turbidity [Show more]Increase in turbidity
EvidenceBarnes & Barnes (1968) found that in high suspended solids and low salinity there was a decrease in the number of eggs per brood of Chthamalus stellatus / Chthamalus montagui. Fecundity in protected areas such as harbours is usually lower, possibly due to increased turbidity (Barnes, 1989). Intolerance is assessed as intermediate. Recoverability is likely to be moderate (see Additional Information section, below.) | Low | Very high | Very Low | Moderate |
Decrease in turbidity [Show more]Decrease in turbidity
EvidenceA decrease in turbidity is likely to lead to an increase in the quantity of flagellates available in the water column. Therefore, the species is assessed as 'tolerant'. | Tolerant | Not relevant | Not sensitive | Not relevant |
Increase in wave exposure [Show more]Increase in wave exposureA change of two ranks on the wave exposure scale (view glossary) e.g., from Exposed to Extremely exposed for a period of one year. Further details EvidenceChthamalus stellatus colonizes exposed rocky shores. An increase in wave exposure is therefore unlikely to affect the species greatly. However, its vertical distribution has been found to increase with increasing wave exposure (Foster, 1971(b)). | Tolerant | Not relevant | Not sensitive | Not relevant |
Decrease in wave exposure [Show more]Decrease in wave exposureA change of two ranks on the wave exposure scale (view glossary) e.g., from Exposed to Extremely exposed for a period of one year. Further details EvidenceChthamalus stellatus colonizes exposed rocky shores. A decrease in wave exposure below 'exposed' is likely to result in replacement of Chthamalus stellatus with Chthamalus montagui, which favours more sheltered environments. A decrease in the level of wave exposure may also cause a shift in the community towards fucoid algae, which prevent barnacle larvae settlement. Intolerance is assessed as intermediate. Recoverability is likely to be moderate (see Additional Information section below). | Intermediate | Moderate | Moderate | High |
Noise [Show more]Noise
EvidenceBarnacles are unlikely to be affected by noise. | Tolerant | Not relevant | Not sensitive | Not relevant |
Visual presence [Show more]Visual presenceBenchmark. The continuous presence for one month of moving objects not naturally found in the marine environment (e.g., boats, machinery, and humans) within the visual envelope of the species or community under consideration. Further details EvidenceBarnacles are unlikely to be affected by visual presence. | Tolerant | Not relevant | Not sensitive | Not relevant |
Abrasion & physical disturbance [Show more]Abrasion & physical disturbanceBenchmark. Force equivalent to a standard scallop dredge landing on or being dragged across the organism. A single event is assumed for assessment. This factor includes mechanical interference, crushing, physical blows against, or rubbing and erosion of the organism or habitat of interest. Where trampling is relevant, the evidence and trampling intensity will be reported in the rationale. Further details. EvidenceCracks and crevices offer protection from some abrasion. Small abrasive forces, such as erosion from suspended sediment, has been noted to cause a decrease in barnacle size (Relilni, 1983). On a larger scale, Gubbay (1983) showed that Chthamalus montagui could withstand a compressive force of 42 newtons (N) and a much lower tensile force of 7.4 N, perhaps equivalent to trampling pressure. It is likely, due to Chthamalus stellatus and Chthamalus montagui being morphologically similar, that these values would be similar for Chthamalus stellatus. However, larger forces are likely to be more harmful to individuals. Therefore, intolerance is assessed as intermediate. Recoverability is likely to be high (see additional information section below). | Intermediate | High | Low | Low |
Displacement [Show more]DisplacementBenchmark. Removal of the organism from the substratum and displacement from its original position onto a suitable substratum. A single event is assumed for assessment. Further details EvidenceOnce the barnacle is fixed in place it is unable to attach again (Crisp, 1955). Intolerance to displacement is therefore assessed as high. Recoverability is likely to be moderate (see Additional Information section below). | High | Moderate | Moderate | High |
Chemical pressures
Use [show more] / [show less] to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Synthetic compound contamination [Show more]Synthetic compound contaminationSensitivity is assessed against the available evidence for the effects of contaminants on the species (or closely related species at low confidence) or community of interest. For example:
The evidence used is stated in the rationale. Where the assessment can be based on a known activity then this is stated. The tolerance to contaminants of species of interest will be included in the rationale when available; together with relevant supporting material. Further details. EvidenceBarnacles have a low resilience to chemicals such as dispersants, dependant on the concentration and type of chemical involved (Holt et al., 1995). They are less intolerant than some species (e.g. Patella vulgata) to dispersants (Southward & Southward, 1978). However, the barnacle population suffered indirectly as a result of the mass mortality of grazers. The resultant bloom of algae, and growth of fucoids, within 6 months, grew over and killed surviving barnacles (Hawkins & Southward, 1992). Therefore, intolerance to synthetic chemicals is assessed as intermediate. Recoverability is likely to be moderate (see Additional Information section, below). | Intermediate | Moderate | Moderate | Very low |
Heavy metal contamination [Show more]Heavy metal contaminationEvidenceBarnacles accumulate heavy metals and store them as insoluble granules. No information is available as to the effects of heavy metals on Chthamalus stellatus, but a larger amount of information was found with respect to a barnacle from the same family, Semibalanus balanoides. It is possible that sensitivities to heavy metals may be similar in both species. Clarke (1947) investigated the intolerance of Semibalanus balanoides to copper, mercury, zinc and silver. He found that 90 percent of barnacles died when held in 0.35 mg/l Cu carbonate for two days. Zinc, mercury and silver killed 90 percent of barnacles in two days at concentrations of 32 mg/l, 1 mg/l and 0.4 mg/l respectively. Pyefinch & Mott (1948) recorded median lethal concentrations of 0.32 mg/l copper and 0.36 mg/l mercury over 24 hours for this species. Barnacles may tolerate fairly high level of heavy metals in nature, for example they are found in Dulas Bay, Anglesey, where copper reaches concentrations of 24.5 µg/l, due to acid mine waste (Foster et al., 1978). Therefore, intolerance is assessed as low. Recoverability is likely to be high (see Additional Information section, below). | Low | High | Low | Very low |
Hydrocarbon contamination [Show more]Hydrocarbon contaminationEvidenceChthamalus stellatus/ Chthamalus montagui have been shown to be relatively unaffected by smothering by oil. Monterosso (1930) showed experimentally that the species can survive complete smothering by petroleum jelly for approximately two months, by respiring anaerobically. Complete smothering caused by the Torrey Canyon oil spill yielded similar results; A few Semibalanus balanoides died, yet Chthamalus stellatus / Chthamalus montagui seemed unaffected, while at Booby's bay more than 90 % had managed to clear an opening in the oil film. On further examination these individuals were found to be in good condition, with no oil present in the gut (Smith, 1968).However, detergents used to clean up the oil lead to a decline in Chthamalus spp. populations. In areas which where large amounts of detergents had been used, there was much greater mortality, and in Kynance cove the population was wiped out completely (Smith, 1968). Therefore, intolerance to hydrocarbons is assessed as low. Recoverability is likely to be high (see Additional Information section, below). | Low | High | Low | Moderate |
Radionuclide contamination [Show more]Radionuclide contaminationEvidenceInsufficientinformation. | No information | Not relevant | No information | Not relevant |
Changes in nutrient levels [Show more]Changes in nutrient levelsEvidenceLittle data exists on the effects of increased nutrients on barnacles. A slight increase in nutrient levels may be beneficial for barnacles by promoting the growth of flagellates. However, Holt et al. (1995) predict that smothering by ephemeral green algae is a possibility under eutrophic conditions. Intolerance to nutrient levels is assessed as intermediate. Recoverability is likely to be high (see Additional Information section, below). | Intermediate | High | Low | Low |
Increase in salinity [Show more]Increase in salinity
EvidenceChthamalids only occur in full salinity water, therefore an increase in salinity is unlikely to exert a noticeable effect. The species is recorded as not intolerant of an increase in salinity. | Tolerant | Not relevant | Not sensitive | Not relevant |
Decrease in salinity [Show more]Decrease in salinity
EvidenceBarnacles are able to acclimatise over a number of days to reduced salinity (Rainbow, 1984), with their closing response is triggered at a lower salinity (Moore & Kitching, 1939). However, the behavioural avoidance is associated with anaerobiosis and low metabolic activity (Barnes et al., 1963). Barnes & Barnes (1965) found that in high suspended solids and low salinity there was a decrease in the number of eggs per brood of Chthamalus stellatus / Chthamalus montagui. If salinities decrease below 21 psu all cirral activity of barnacles normally associated with full salinity waters, ceases (Foster, 1971). Therefore, intolerance to a decrease in salinity is assessed as high. Recoverability is likely to be high (see Additional Information section, below). | High | High | Moderate | Moderate |
Changes in oxygenation [Show more]Changes in oxygenationBenchmark. Exposure to a dissolved oxygen concentration of 2 mg/l for one week. Further details. EvidenceSouthward (1955) conducted experiments on the relationship of cirral activity in Chthamalus stellatus / Chthamalus montagui, connected with feeding and respiration, to decreased oxygenation, by passing nitrogen through the water at 6 ml per minute at 13 °C. He found that in all cases a decrease in oxygen concentration lead to a decrease in cirral activity and that, after 15 minutes, the mean cirral beat had decreased from 3.1 to 2.9 beats per second. After 30 minutes exposure, cirral beat had completely ceased and the barnacle remained inactive. It was further observed that the scuta and terga remained slightly open with the cirri often protruding.Barnacles have to obtain oxygen from the water through their cirri including by cirral beating in still water. Since cirri stop beating in response to lowered oxygen levels, it seems likely that intolerance will be high. Therefore, intolerance to oxygen levels is assessed as high. Recoverability is likely to be high (see Additional information section, below). | High | High | Moderate | Moderate |
Biological pressures
Use [show more] / [show less] to open/close text displayed
| Intolerance | Recoverability | Sensitivity | Evidence / Confidence | |
Introduction of microbial pathogens/parasites [Show more]Introduction of microbial pathogens/parasitesBenchmark. Sensitivity can only be assessed relative to a known, named disease, likely to cause partial loss of a species population or community. Further details. EvidenceHealy (1986, in O'Riordan et al., 1992) observed the crustacean parasite, Hemioniscus balani, in Chthamalus stellatus and Chthamalus montagui in Ireland, although it was never present in Lough Hyne. However, Southward & Crisp (1954) found that although it attacks and sterilises Semibalanus balanoides individuals, it does not attack Chthamalids, at least not in the British Isles. Therefore, intolerance to microbial pathogens / parasites is assessed as intermediate. Recoverability is likely to be high (see Additional Information section, below). | Intermediate | High | Low | Low |
Introduction of non-native species [Show more]Introduction of non-native speciesSensitivity assessed against the likely effect of the introduction of alien or non-native species in Britain or Ireland. Further details. EvidenceThe Australasian barnacle Elminius modestus was introduced to British waters on ships during the second world war. Although the species does well in estuaries and bays, where it can displace Semibalanus balanoides and Chthamalus montagui, Chthamalus stellatus generally occurs in more exposed locations where Elminius modestus is likely to be less abundant or absent. Therefore, intolerance to non-native species is assessed as low. Recoverability is likely to be high. | Low | High | Low | Moderate |
Extraction of this species [Show more]Extraction of this speciesBenchmark. Extraction removes 50% of the species or community from the area under consideration. Sensitivity will be assessed as 'intermediate'. The habitat remains intact or recovers rapidly. Any effects of the extraction process on the habitat itself are addressed under other factors, e.g. displacement, abrasion and physical disturbance, and substratum loss. Further details. EvidenceNot relevant. | Not relevant | Not relevant | Not relevant | Not relevant |
Extraction of other species [Show more]Extraction of other speciesBenchmark. A species that is a required host or prey for the species under consideration (and assuming that no alternative host exists) or a keystone species in a biotope is removed. Any effects of the extraction process on the habitat itself are addressed under other factors, e.g. displacement, abrasion and physical disturbance, and substratum loss. Further details. EvidenceCollection of intertidal algae may damage barnacles by abrasion from trampling. Intolerance is assessed as low. Recoverability is likely to be high (see Additional Information section, below). | Low | High | Low | Low |
Additional information
Southward & Crisp (1954) suggested that Chthamalus stellatus is able to breed in its first year after 9 to 10 months of settlement. Throughout the breeding season most individuals produce several broods (Burrows et al., 1992; O'Riordan et al., 1992), with a small percentage of the population remaining reproductively active throughout the year (O'Riordan et al., 1995)Importance review
Policy/legislation
- no data -
Status
| National (GB) importance | - | Global red list (IUCN) category | - |
Non-native
| Parameter | Data |
|---|---|
| Native | Native |
| Origin | - |
| Date Arrived | - |
Importance information
-none-Bibliography
Anderson, D.T. & Southward, A.J., 1987. Cirrial activity of barnacles. In Barnacle biology, (ed. A.J. Southward), pp. 135-174. Rotterdam: A.A. Balkema. [Crustacean Issues no. 5.]
Anderson, D.T., 1994. Barnacles. Structure, function, development and evolution. Melbourne: Chapman & Hall.
Barnes, H. & Barnes, M., 1965. Egg size, nauplius size, and their variation with local, geographical and specific factors in some common cirripedes. Journal of Animal Ecology, 34, 391-402.
Barnes, H. & Barnes, M., 1968. Egg numbers, metabolic efficiency and egg production and fecundity; local and regional variations in a number of common cirripedes. Journal of Experimental Marine Biology and Ecology, 2, 135-153.
Barnes, H., 1953. The effect of lowered salinity on some barnacle nauplii. Journal of Animal Ecology, 22, 328-330.
Barnes, H., Finlayson, D.M. & Piatigorsky, J., 1963. The effect of desiccation and anaerobic conditions on the behaviour, survival and general metabolism of three common cirripedes. Journal of Animal Ecology, 32, 233-252.
Barnes, M., 1989. Egg production in Cirripedia. Oceanography and Marine Biology: an Annual Review, 27, 91-166.
Barnes, M., 1992. The reproductive periods and condition of the penis in several species of common cirripedes. Oceanography and Marine Biology: an Annual Review, 30, 483-525.
Bassindale, R., 1964. British Barnacles. London: The Linnean Society of London.[Synopses of the British Fauna, no. 14.]
Benedetti-Cecchi, L., Acunto, S., Bulleri, F. & Cinelli, F., 2000. Population ecology of the barnacle Chthamalus stellatus in the northwest Mediterranean. Marine Ecology Progress Series, 198, 157-170.
Bokn, T.L., Moy, F.E. & Murray, S.N., 1993. Long-term effects of the water-accommodated fraction (WAF) of diesel oil on rocky shore populations maintained in experimental mesocosms. Botanica Marina, 36 (4), 313-319. DOI https://doi.org./10.1515/botm.1993.36.4.313
Bourget, E., 1977. Shell structure in sessile barnacles. Le Naturaliste Canadien, 104, 281-323.
Burrows, M.T., 1988. The comparative biology of Chthamalus Stellatus (Poli) and Chthamalus montagui Southward. PhD thesis, University of Manchester. 318pp.,
Burrows, M.T., Hawkins, S.J. & Southward, A.J., 1992. A comparison of reproduction in co-occurring chthamalid barnacles, Chthamalus stellatus (Poli) and Chthamalus montagui Southward. Journal of Experimental Marine Biology and Ecology, 160, 229-249.
Burrows, M.T., Hawkins, S.J. & Southward, A.J., 1999. Larval development of the intertidal barnacles Chthamalus stellatus and Chthamalus montagui. Journal of the Marine Biological Association of the United Kingdom, 79, 93-101.
Clarke, G.L., 1947. Poisoning and recovery in barnacles and mussels. Biological Bulletin, Marine Biological Laboratory, Woods Hole, 92, 73-91.
Crisp, D.J. & Barnes, H., 1954. The orientation and distribution of barnacles at settlement with particular reference to surface contour. Journal of Animal Ecology, 23, 142-162.
Crisp, D.J. & Bourget, E., 1985. Growth in Barnacles. Advances in Marine Biology, 22, 199-244.
Crisp, D.J. & Patel, B.S., 1960. The moulting cycle in Balanus balanoides (L.). Biological Bulletin, Marine Biological Laboratory, Woods Hole, 118, 31-47.
Crisp, D.J. (ed.), 1964. The effects of the severe winter of 1962-63 on marine life in Britain. Journal of Animal Ecology, 33, 165-210.
Crisp, D.J., 1950. Breeding and distribution of Chthamalus stellatus. Nature (London), 166, 311-312.
Crisp, D.J., 1955. The behaviour of barnacle cyprids in relation to water movement over a surface. Journal of Experimental Biology, 32, 569-590.
Crisp, D.J., 1974. Factors influencing the settlement of marine invertebrate larvae. In Chemoreception in Marine Organisms, Chapter 5 (ed. P.T. Grant & A.M. Mackie), pp. 177-265. London: Academic Press.
Crisp, D.J., Southward, A.J. & Southward, E.C., 1981. On the distribution of the intertidal barnacles Chthamalus stellatus, Chthamalus montagui and Euraphia depressa. Journal of the Marine Biological Association of the United Kingdom, 61, 359-380.
Fish, J.D. & Fish, S., 1996. A student's guide to the seashore. Cambridge: Cambridge University Press.
Foster, B.A., 1971a. Desiccation as a factor in the intertidal zonation of barnacles. Marine Biology, 8, 12-29.
Foster, B.A., 1971b. On the determinants of the upper limit of intertidal distribution of barnacles. Journal of Animal Ecology, 40, 33-48.
Foster, P., Hunt, D.T.E. & Morris, A.W., 1978. Metals in an acid mine stream and estuary. Science of the Total Environment, 9, 75-86.
Gubbay, S., 1983. Compressive and adhesive strengths of a variety of British barnacles. Journal of the Marine Biological Association of the United Kingdom, 63, 541-555.
Hawkins, S.J. & Hartnoll, R.G., 1982. Settlement patterns of Semibalanus balanoides in the Isle of Man (1977-1981). Journal of Experimental Marine Biology and Ecology, 62, 271-283.
Hawkins, S.J. & Southward, A.J., 1992. The Torrey Canyon oil spill: recovery of rocky shore communities. In Restoring the Nations Marine Environment, (ed. G.W. Thorpe), Chapter 13, pp. 583-631. Maryland, USA: Maryland Sea Grant College.
Hayward, P., Nelson-Smith, T. & Shields, C. 1996. Collins pocket guide. Sea shore of Britain and northern Europe. London: HarperCollins.
Hayward, P.J. & Ryland, J.S. (ed.) 1995b. Handbook of the marine fauna of North-West Europe. Oxford: Oxford University Press.
Hines, A.H., 1978. Reproduction in three species of intertidal barnacle from central California. Biological Bulletin, Marine Biological Laboratory, Woods Hole, 154, 262-281.
Howson, C.M. & Picton, B.E., 1997. The species directory of the marine fauna and flora of the British Isles and surrounding seas. Belfast: Ulster Museum. [Ulster Museum publication, no. 276.]
Kendall, M.A. & Bedford, M.L., 1987. Reproduction and recruitment of the barnacle Chthamalus montagui at Aberystwyth (mid-Wales). Marine Ecology Progress Series, 38, 305-308.
Lewis, J.R., 1964. The Ecology of Rocky Shores. London: English Universities Press.
Mathieson, A.C., Neefus, C.D. & Penniman, C.E., 1983. Benthic ecology in an estuarine tidal rapid. Botanica Marina, 26, 213-230.
Monterosso, B., 1930. Studi cirripedologici. VI. Sul comportamento di Chthamalus stellatus in diverse condizioni sperimentali. Atti Accad. Naz. Lincei Rc., 9, 501-504.
Moore, H.B. & Kitching, J.A., 1939. The biology of Chthamalus stellatus (Poli). Journal of the Marine Biological Association of the United Kingdom, 23, 521-541.
Moore, P.G. & Seed, R. (ed.), 1985. The Ecology of Rocky Coasts. London: Hodder and Stoughton Publ.
Mortlock, A.M., Fitzsimons, J.T.R. & Kerkaut, G.A., 1984. The effects of farnesol on the late stage nauplius and free swimming cypris larvae of Elminius modestus. Comparative Biochemistry and Physiology, 78A, 345-357.
O'Riordan, R.M., Myers, A.A. & Cross, T.F., 1992. Brooding in the intertidal barnacles Chthamalus stellatus (Poli) and Chthamalus montagui (Southward) in south-western Ireland. Journal of Experimental Marine Biology and Ecology, 164, 135-145.
O'Riordan, R.M., Myers, A.A. & Cross, T.F., 1995. The reproductive cycles of Chthamalus stellatus (Poli) and Chthamalus montagui (Southward) in south-western Ireland. Journal of Experimental Marine Biology and Ecology, 190, 17-38.
O'Riordan, R.M., Myers, A.A., McGrath, D., Delany, J. & Power, A-M., 1999. The sizes at settlement in natural populations of the cyprids of Chthamalus montagui and Chthamalus stellatus. Journal of the Marine Biological Association of the United Kingdom, 79, 365-366.
Pannacciulli, F.G. & Relini, G., 2000. The vertical distribution of Chthamalus montagui and Chthamalus stellatus (Crustacea, Cirripedia) in two areas of the NW Mediterranean Sea. Hydrobiologia, 426, 105-112.
Patel, B. & Crisp, D. J., 1960. The influence of temperature on the breeding and the moulting activities of some warm-water species of operculate barnacles. Journal of the Marine Biological Association of the United Kingdom, 36, 667-680.
Patel, B. & Crisp, D.J., 1960. Rates of development of the embryos of several species of barnacles. Physiology and Zoology, 33, 104-119.
Pyefinch, K.A. & Mott, J.C., 1948. The sensitivity of barnacles and their larvae to copper and mercury. Journal of Experimental Biology, 25, 276-298.
Raffaelli, D.G. & Hawkins, S.J., 1999. Intertidal Ecology 2nd edn.. London: Kluwer Academic Publishers.
Rainbow, P.S., 1984. An introduction to the biology of British littoral barnacles. Field Studies, 6, 1-51.
Relini, G., 1983. Remarks on the ecology of Chthamalids in the Ligurian Sea. Rapp. P.-V. Reun. Ciesm., 28, 273-275.
Smith, J.E. (ed.), 1968. 'Torrey Canyon'. Pollution and marine life. Cambridge: Cambridge University Press.
Southward, A., 1978. Marine life and Amoco Cadiz. New Scientist, 79, 174-176
Southward, A.J. & Crisp, D.J., 1954. The distribution of certain intertidal animals around the Irish coast. Proceedings of the Royal Irish Academy, 57B, 1-29.
Southward, A.J. & Crisp, D.J., 1956. Fluctuations in the distribution and abundance of intertidal barnacles. Journal of the Marine Biological Association of the United Kingdom, 35, 211-229.
Southward, A.J. & Crisp, D.J., 1965. Activity rhythms of barnacles in relation to respiration and feeding. Journal of the Marine Biological Association of the United Kingdom, 45, 161-185.
Southward, A.J. & Southward, E.C., 1978. Recolonisation of rocky shores in Cornwall after use of toxic dispersants to clean up the Torrey Canyon spill. Journal of the Fisheries Research Board of Canada, 35, 682-706.
Southward, A.J., 1955. On the behaviour of barnacles. I. The relation of cirral and other activities to temperature. Journal of the Marine Biological Association of the United Kingdom, 34, 403-432.
Southward, A.J., 1955b. On the behaviour of barnacles II. The influence of habitat and tidal level on cirral activity. Journal of the Marine Biological Association of the United Kingdom, 34, 423-433.
Southward, A.J., 1958. Note on the temperature tolerances of some intertidal animals in relation to environmental temperatures and geographical distribution. Journal of the Marine Biological Association of the United Kingdom, 37, 49-56.
Southward, A.J., 1964. Limpet grazing and the control of vegetation on rocky shores. In Grazing in Terrestrial and Marine Environments, British Ecological Society Symposium No. 4 (ed. D.J. Crisp), 265-273.
Southward, A.J., 1964b. The relationship between temperature and rhythmic cirral activity in some Cirripedia considered in connection with their geographical distribution. Helgolander Wissenschaftliche Meeresuntersuchungen, 10, 391-403.
Southward, A.J., 1967. Recent changes in abundance of intertidal barnacle in south-west England: a possible effect of climatic deterioration. Journal of the Marine Biological Association of the United Kingdom, 47, 81-85.
Southward, A.J., 1976. On the taxonomic status and distribution of Chthamalus stellatus (Cirripedia) in the north-eastern Atlantic region: with a key to the common intertidal barnacles of Britain. Journal of the Marine Biological Association of the United Kingdom, 56, 1007-1028.
Southward, A.J., 1991. Forty years of changes in species composition and population density of barnacles on a rocky shore near Plymouth. Journal of the Marine Biological Association of the United Kingdom, 71, 495-513.
Stubbings, H.G., 1975. Balanus balanoides. Liverpool: Liverpool University Press.
Tighe-Ford, D.J., 1977. Effects of juvenile hormone analogues on larval metamorphosis in the barnacle Elminius modestus Darwin. Journal of Experimental Marine Biology and Ecology, 26, 163-176.
Walker, G., 1995. Larval settlement: Historical and future perspectives. In New Frontiers in Barnacle Evolution, (ed. F.R. Schram & J.T. Hoeg). Rotterdam: A.A. Balkema. [Crustacean Issues 10]
Willemsen, P.R., Overbeke, K. & Suurmond, A., 1998. Repetitive testing of TBTO, Sea-nine 211 and farnesol using Balanus amphitrite (Darwin) cypris larvae: variability in larval sensitivity. Biofouling, 12, 133-147.
Wu, R.S.S., Lam, P.K.S. & Zhou, B.S., 1997. Effects of two oil dispersants on phototaxis and swimming behaviour of barnacle larvae. Hydrobiologia, 352, 9-16.
Datasets
Centre for Environmental Data and Recording, 2018. Ulster Museum Marine Surveys of Northern Ireland Coastal Waters. Occurrence dataset https://www.nmni.com/CEDaR/CEDaR-Centre-for-Environmental-Data-and-Recording.aspx accessed via NBNAtlas.org on 2018-09-25.
Cofnod – North Wales Environmental Information Service, 2018. Miscellaneous records held on the Cofnod database. Occurrence dataset: https://doi.org/10.15468/hcgqsi accessed via GBIF.org on 2018-09-25.
Fenwick, 2018. Aphotomarine. Occurrence dataset http://www.aphotomarine.com/index.html Accessed via NBNAtlas.org on 2018-10-01
National Trust, 2017. National Trust Species Records. Occurrence dataset: https://doi.org/10.15468/opc6g1 accessed via GBIF.org on 2018-10-01.
NBN (National Biodiversity Network) Atlas. Available from: https://www.nbnatlas.org.
OBIS (Ocean Biodiversity Information System), 2024. Global map of species distribution using gridded data. Available from: Ocean Biogeographic Information System. www.iobis.org. Accessed: 2024-05-12
Outer Hebrides Biological Recording, 2018. Invertebrates (except insects), Outer Hebrides. Occurrence dataset: https://doi.org/10.15468/hpavud accessed via GBIF.org on 2018-10-01.
South East Wales Biodiversity Records Centre, 2018. SEWBReC Myriapods, Isopods, and allied species (South East Wales). Occurrence dataset: https://doi.org/10.15468/rvxsqs accessed via GBIF.org on 2018-10-02.
South East Wales Biodiversity Records Centre, 2018. Dr Mary Gillham Archive Project. Occurance dataset: http://www.sewbrec.org.uk/ accessed via NBNAtlas.org on 2018-10-02
Citation
This review can be cited as:
Last Updated: 28/01/2002